Online Database of Chemicals from Around the World
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zhejiang Metallurgical Research Institute Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pgmcatalyst.com | |||
 | +86 (571) 8817-8133 +86 (578) 814-5985 | |||
 | +86 (578) 814-5986 | |||
 | xk@pgmcatalyst.com scm@pgmcatalyst.com ylq@pgmcatalyst.com | |||
| Chemical manufacturer since 1958 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Larui Innovation Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmaluxury.com | |||
 | +86 (25) 8372-6959 | |||
 | +86 (25) 8666-0986 | |||
 | goodsalejs@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Auspai Pty Lty | Australia | |||
|---|---|---|---|---|
 | www.auspai.com.au | |||
 | +61 (426) 886-972 | |||
 | frankquy@gmail.com | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Henan Tianfu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianfuchem.com | |||
 | +86 (371) 5517-0693 | |||
 | +86 (371) 5517-0975 | |||
 | leon@tianfuchem.com | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Beijing Guofang Dingsheng Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gfdschem.com | |||
 | +86 (10) 8904-9254 5909-2859 +86 18610472091 +86 18031679351 | |||
 | +86 (10) 5909-2859 | |||
 | gfdskj@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Kangchi Chemical Industry (shenzhen) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.konchie.com | |||
 | +86 (755) 8257-4253 | |||
 | +86 (755) 3315-1032 | |||
 | kangchi@konchie.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Synthesis With Catalysts Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.synthesiswithcatalysts.com | |||
 | +91 (120) 431-9102 | |||
 | +91 (120) 431-9102 | |||
 | sales1@synthesiswithcatalysts.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Wuhan Jdchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jdchem.com.cn | |||
 | +86 18271874579 | |||
 | sales@JDchem.com.cn | |||
 | QQ Chat | |||
 | WeChat: 18271874579 | |||
 | WhatsApp:08618271874579 | |||
| Chemical manufacturer since 2022 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wu Han Taiyu Kaishang Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taiyuchem.com | |||
 | +86 13657232893 | |||
 | 13657232893@163.com | |||
 | WhatsApp:+86 13657232893 | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2025 | ||||
| MCAT GmbH | Germany | |||
|---|---|---|---|---|
 | www.mcat.de | |||
 | +49 (7531) 939-097 | |||
 | +49 (7531) 939-098 | |||
 | info@mcat.de | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| W.C.Heraeus GmbH | Germany | |||
|---|---|---|---|---|
 | www.heraeus-chemicalproducts.com | |||
 | +49 (6181) 355-707 | |||
 | +49 (6181) 355-914 | |||
 | info@heraeus.com | |||
| Chemical manufacturer | ||||
| Metalor Technologies Group | China | |||
|---|---|---|---|---|
 | www.metalor.com | |||
 | +86 (512) 6593-6181 +86 (21) 5442-3060 | |||
 | +86 (512) 6593-6171 / +86 (21) 5442-0790 | |||
 | hr.cn@metalor.com | |||
| Chemical manufacturer | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Colonial Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.colonialmetals.com | |||
 | +1 (410) 398-7200 (800) 962-1537 | |||
 | +1 (410) 398-2918 | |||
 | sales@colonialmetals.com | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic palladium |
|---|---|
| Name | Bis(triphenylphosphine)palladium(II) chloride |
| Synonyms | Dichlorobis(triphenylphosphine)palladium(II); Palladium(II)bis(triphenylphosphine) dichloride |
| Molecular Structure | 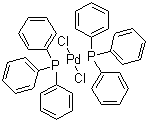 |
| Molecular Formula | C36H30Cl2P2Pd |
| Molecular Weight | 701.91 |
| CAS Registry Number | 13965-03-2 |
| EC Number | 237-744-2 |
| SMILES | C1=CC=C(C=C1)P(C2=CC=CC=C2)C3=CC=CC=C3.C1=CC=C(C=C1)P(C2=CC=CC=C2)C3=CC=CC=C3.Cl[Pd]Cl |
| Solubility | Soluble (chloroform, hexane toluene) |
|---|---|
| Melting point | 310 °C (decomp.) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H413 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P273-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Bis(triphenylphosphine)palladium(II) chloride, often abbreviated as Pd(PPh3)2Cl2, is a widely studied organometallic compound. The synthesis of bis(triphenylphosphine)palladium(II) chloride was first reported in the mid-20th century when chemists were exploring the coordination chemistry of palladium with phosphine ligands. The compound is typically prepared by reacting palladium(II) chloride with triphenylphosphine in a suitable solvent under controlled conditions. The reaction forms a deep red crystalline solid characterized by unique catalytic and organic transformation properties. The chemical formula of bis(triphenylphosphine)palladium(II) chloride is Pd(PPh₃)₂Cl₂. It consists of a palladium ion (Pd²⁺) coordinated to two triphenylphosphine (PPh₃) ligands and two chloride ions (Cl⁻) in a square planar geometry. This molecular structure confers stability and reactivity, making it an effective catalyst in a variety of reactions. The compound is slightly soluble in polar solvents such as acetone and dichloromethane, but more soluble in nonpolar solvents such as benzene and toluene. It is stable under ambient conditions but is sensitive to air and moisture and needs to be handled under an inert atmosphere for preservation. Bis(triphenylphosphine)palladium(II) chloride is valued for its catalytic properties, especially in palladium-catalyzed cross-coupling reactions. The chloride ion can be replaced by other ligands, which affects the reactivity and selectivity of the compound in different catalytic processes. Pd(PPh₃)₂Cl₂ is an important catalyst in the Suzuki-Miyaura coupling reaction, which forms aryl-aryl, aryl-vinyl, and aryl-alkyl bonds under mild conditions. This reaction is essential for building complex molecular structures in drug synthesis, material science, and agrochemical production. It catalyzes the Heck reaction, which promotes the coupling of aryl halides with alkenes to form substituted alkenes. This transformation is important for modifying natural products and designing functional materials with specific electronic and structural properties. Pd(PPh₃)₂Cl₂ is used in the Mizoroki-Heck reaction to couple aryl halides with alkynes or alkenes in the presence of carbon monoxide (CO). The process leads to the synthesis of α,β-unsaturated carbonyl compounds, which are key intermediates in the production of pharmaceuticals and fine chemicals. It is used as a catalyst for hydrogenation reactions, promoting the reduction of alkenes, alkynes, and carbonyl compounds to the corresponding saturated derivatives. This catalytic ability is crucial in the production of chiral compounds for pharmaceuticals and in industrial processes for the conversion of unsaturated hydrocarbons. Bis(triphenylphosphine)palladium(II) chloride and its derivatives are immobilized on solid supports or nanoparticle surfaces for heterogeneous catalysis. This application improves the recyclability, stability, and efficiency of the catalyst in large-scale industrial processes. References 2024. Synthetic studies on the tetrasubstituted D-ring of cystobactamids lead to potent terephthalic acid antibiotics. Communications Chemistry. DOI: 10.1038/s42004-024-01337-6 2015. Palladium-Catalyzed Carbonylative Synthesis of Six-Membered Heterocycles from Aryl Halides. Transition Metal Catalyzed Carbonylative Synthesis of Heterocycles. DOI: 10.1007/7081_2015_150 2012. Metalation Reactions of Isoxazoles and Benzisoxazoles. Metalation of Azoles and Related Five-Membered Ring Heterocycles. DOI: 10.1007/7081_2012_80 |
| Market Analysis Reports |