Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | Bis(triphenylphosphine)cuprous nitrate |
| Molecular Structure | 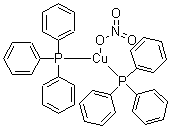 |
| Molecular Formula | C36H30CuP2.NO3 |
| Molecular Weight | 650.12 |
| CAS Registry Number | 106678-35-7 |
| EC Number | 631-247-8 |
| SMILES | C1=CC=C(C=C1)P(C2=CC=CC=C2)C3=CC=CC=C3.C1=CC=C(C=C1)P(C2=CC=CC=C2)C3=CC=CC=C3.[N+](=O)([O-])[O-].[Cu+] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Bis(triphenylphosphine)cuprous nitrate, often abbreviated as [Cu(PPh₃)₂NO₃] or CuI(PPh₃)₂NO₃, is a noteworthy complex in coordination chemistry, particularly valued for its role in various catalytic applications. This compound combines a cuprous ion with two triphenylphosphine ligands and a nitrate anion, forming a versatile and functional entity. The discovery of bis(triphenylphosphine)cuprous nitrate can be traced back to the study of copper(I) complexes and their properties. The preparation of this complex typically involves reacting copper(I) nitrate with triphenylphosphine in a suitable solvent. The resulting compound is a solid that is usually characterized by techniques such as X-ray crystallography, which confirms its molecular structure. In terms of its applications, bis(triphenylphosphine)cuprous nitrate is primarily utilized in catalysis. Copper(I) complexes, including this one, are known for their ability to facilitate various organic transformations. The presence of triphenylphosphine ligands provides steric and electronic effects that influence the reactivity and selectivity of the copper center. This makes the complex particularly useful in catalyzing reactions such as cross-coupling, where copper(I) species can act as efficient catalysts. One of the notable applications of bis(triphenylphosphine)cuprous nitrate is in the field of organic synthesis, specifically in the coupling reactions. These include the Ullmann reaction and the Goldberg reaction, where the complex serves as a catalyst to facilitate the formation of carbon-carbon bonds. The use of copper(I) complexes like this one has become increasingly popular due to their ability to provide effective and cost-efficient alternatives to palladium-based catalysts. Additionally, bis(triphenylphosphine)cuprous nitrate has found applications in the synthesis of complex organic molecules and in the preparation of pharmaceuticals. Its ability to mediate various bond-forming reactions makes it a valuable tool for chemists looking to construct intricate structures in a straightforward manner. The complex is also of interest in the study of copper(I) chemistry and its behavior in coordination environments. The combination of triphenylphosphine ligands with a cuprous ion provides a model system for exploring the electronic and steric effects in copper(I) coordination complexes. This contributes to a broader understanding of copper's role in catalysis and other chemical processes. In summary, bis(triphenylphosphine)cuprous nitrate is a significant compound in coordination chemistry with diverse applications, particularly in catalysis. Its role in facilitating organic transformations and its utility in the synthesis of complex molecules underscore its importance in both research and industrial applications. References 2022. C—H Nitration with Silver(I) Nitrate and Silver(I) Nitrite. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-238-00096 1985. C36H20 - C36H31 (4745 - 4785). Landolt-Börnstein - Group III Condensed Matter. DOI: 10.1007/10201616_39 |
| Market Analysis Reports |