Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic palladium |
|---|---|
| Name | rac-BINAP-Pd-G3 |
| Synonyms | 2'-(Amino-?N)[1,1'-biphenyl]-2-yl-?C][[2'-(diphenylphosphino)[1,1'-binaphthalen]-2-yl]diphenylphosphine-?P](methanesulfonato-?O)palladium |
| Molecular Structure | 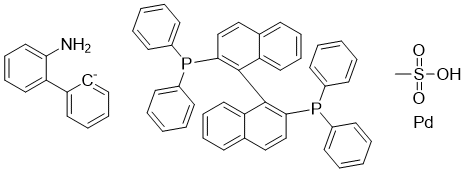 |
| Molecular Formula | C57H46NO3P2PdS- |
| Molecular Weight | 993.41 |
| CAS Registry Number | 2151915-22-7 |
| SMILES | CS(=O)(=O)O.C1=CC=C(C=C1)P(C2=CC=CC=C2)C3=C(C4=CC=CC=C4C=C3)C5=C(C=CC6=CC=CC=C65)P(C7=CC=CC=C7)C8=CC=CC=C8.C1=CC=C([C-]=C1)C2=CC=CC=C2N.[Pd] |
| Melting Point | 175-180 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335-H412 Details |
| Safety Statements | P261-P264-P271-P273-P280-P302+P352-P304+P340+P312-P305+P351+P338-P332+P313-P337+P313-P403+P233-P405-P501 Details |
| SDS | Available |
|
Rac-BINAP-Pd-G3 is a notable chemical compound in the realm of organometallic chemistry and catalysis. This complex features a palladium center coordinated with racemic 2,2'-bis(diphenylphosphino)-1,1'-binaphthyl (BINAP) ligands. The "G3" designation indicates it is a third-generation variant, reflecting advancements in its design and application. The discovery of rac-BINAP-Pd-G3 was motivated by the need for highly effective and selective catalysts for various organic transformations. BINAP is well-known for its ability to stabilize palladium centers and influence their reactivity due to its bidentate nature and chiral framework. The racemic version of BINAP used in rac-BINAP-Pd-G3 provides a broader range of applications by combining both enantiomers of BINAP, which can influence the catalyst's performance in asymmetric synthesis. Rac-BINAP-Pd-G3 is primarily applied in cross-coupling reactions, such as Suzuki-Miyaura and Heck reactions. These reactions are crucial for the formation of carbon-carbon bonds and are extensively used in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. The effectiveness of rac-BINAP-Pd-G3 in these processes is attributed to the optimal electronic and steric environment provided by the BINAP ligand, which enhances the palladium catalyst's ability to facilitate bond formation with high selectivity and efficiency. Additionally, the third-generation designation of rac-BINAP-Pd-G3 suggests improvements in the ligand design, which contribute to greater stability and higher activity compared to earlier generations. These enhancements are achieved through modifications in the ligand structure, optimizing the catalyst's performance and making it suitable for more demanding synthetic applications. The compound also finds applications in other catalytic processes, including polymerizations and various transformations involving palladium catalysis. Its versatility and efficiency make it a valuable tool in both academic research and industrial chemistry, where precision and effectiveness are crucial. In summary, rac-BINAP-Pd-G3 represents a significant advancement in palladium-catalyzed chemistry. Its development has provided researchers with a powerful catalyst for a wide range of organic reactions, contributing to the progress in chemical synthesis and material science. References none |
| Market Analysis Reports |