Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic palladium |
|---|---|
| Name | meCgPPh Pd G3 |
| Synonyms | [(1,3,5,7-Tetramethyl-6-phenyl-2,4,6-trioxa-6-phosphaadamantane)-2-(2'-amino-1,1'-biphenyl)]palladium(II) methanesulfonate |
| Molecular Structure | 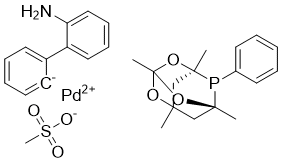 |
| Molecular Formula | C29H34NO6PPdS |
| Molecular Weight | 662.04 |
| CAS Registry Number | 2230788-58-4 |
| SMILES | C[C@]12CC3(C[C@](P1C4=CC=CC=C4)(OC(O2)(O3)C)C)C.CS(=O)(=O)[O-].C1=CC=C([C-]=C1)C2=CC=CC=C2N.[Pd+2] |
| Melting Point | 130-136 °C (decomp.) |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H312-H332 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P330-P363-P501 Details |
| SDS | Available |
|
MeCgPPh Pd G3 is a notable chemical compound in the field of organometallic chemistry, particularly valued for its role as a palladium catalyst in various synthetic reactions. This compound consists of a palladium center coordinated with the MeCgPPh ligand, a specialized phosphine ligand known for its significant impact on catalytic performance. The discovery of MeCgPPh Pd G3 emerged from efforts to develop more efficient and selective catalysts for organic transformations. The ligand MeCgPPh, characterized by its unique structure, was designed to enhance the stability and reactivity of palladium complexes. The "G3" designation indicates that this is a third-generation version of the ligand, incorporating design improvements that optimize its catalytic properties. MeCgPPh Pd G3 is primarily used in cross-coupling reactions, such as the Suzuki-Miyaura and Heck reactions, which are essential for forming carbon-carbon bonds. These reactions are fundamental in the synthesis of complex organic molecules, including pharmaceuticals, agrochemicals, and advanced materials. The effectiveness of MeCgPPh Pd G3 in these reactions is attributed to the ligand's ability to stabilize the palladium center and facilitate reactions with high selectivity and efficiency. The third-generation design of MeCgPPh Pd G3 reflects advancements in ligand development, aimed at enhancing the catalyst's performance under a wider range of conditions and improving its tolerance to various functional groups. This results in increased reaction yields and selectivity, making MeCgPPh Pd G3 a valuable tool in both academic research and industrial applications. In addition to its role in cross-coupling reactions, MeCgPPh Pd G3 is also utilized in other palladium-catalyzed processes, including polymerizations and various coupling reactions. Its versatility and efficiency make it a preferred choice for researchers and industrial chemists working on complex synthetic challenges. Overall, MeCgPPh Pd G3 represents a significant advancement in palladium-catalyzed chemistry. Its development provides a powerful catalyst for a wide range of organic reactions, contributing to progress in chemical synthesis and material science. References none |
| Market Analysis Reports |