Online Database of Chemicals from Around the World
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| 2A Pharmachem USA | USA | |||
|---|---|---|---|---|
 | www.2apharmachem.com | |||
 | +1 (630) 322-8887 | |||
 | +1 (630) 322-8885 | |||
 | sales@2abiotech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chsdpharma.com | |||
 | +86-531-58897029 | |||
 | ericqiao@jnchsd.com | |||
 | QQ Chat | |||
 | WeChat: 15550412551 | |||
 | WhatsApp:15550412551 | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Cambrex Corporation | USA | |||
|---|---|---|---|---|
 | www.cambrex.com | |||
 | +1 (201) 804-3000 | |||
 | +1 (201) 804-9852 | |||
 | info@cambrex.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Anesthetic agents >> Local anesthetics |
|---|---|
| Name | Bupivacaine |
| Synonyms | 1-Butyl-N-(2,6-dimethylphenyl)piperidine-2-carboxamide |
| Molecular Structure | 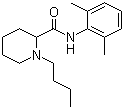 |
| Molecular Formula | C18H28N2O |
| Molecular Weight | 288.43 |
| Protein Sequence | X |
| CAS Registry Number | 2180-92-9 |
| EC Number | 218-553-3 |
| SMILES | CCCCN1CCCCC1C(=O)NC2=C(C=CC=C2C)C |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 423.4±45.0 °C 760 mmHg (Calc.)* |
| Flash point | 209.9±28.7 °C (Calc.)* |
| Index of refraction | 1.547 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H310-H330 Details | ||||||||||||||||||||||||
| Safety Statements | P260-P262-P264-P270-P271-P280-P284-P301+P316-P302+P352-P304+P340-P316-P320-P321-P330-P361+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||
|
Bupivacaine is a widely used amide-type local anesthetic known for its long duration of action and potent anesthetic effects. It functions by blocking voltage-gated sodium channels in nerve cell membranes, which prevents the initiation and propagation of nerve impulses, leading to a reversible loss of sensation in the area where it is administered. Bupivacaine is commonly employed for regional anesthesia techniques, including epidural, spinal, peripheral nerve blocks, and infiltration anesthesia. Bupivacaine was first synthesized in the 1950s and introduced into clinical practice shortly thereafter as an improvement over earlier local anesthetics. Its development aimed to provide prolonged anesthesia with good potency and manageable toxicity. Bupivacaine’s chemical structure is characterized by a butyl group attached to the nitrogen of the amide linkage, which contributes to its lipophilicity and duration of action. The hydrochloride salt of bupivacaine is the form most commonly used in medical settings. It is available in various concentrations and formulations, including solutions for injection, often combined with vasoconstrictors such as epinephrine to prolong anesthetic effects and reduce systemic absorption. The drug exhibits a relatively slow onset compared to agents like lidocaine but provides anesthesia lasting from two to eight hours depending on the dose, concentration, and site of administration. Clinically, bupivacaine is preferred in procedures that require extended anesthesia, such as major surgeries, labor epidurals, and postoperative pain management. Its potency and prolonged effect make it particularly useful for continuous infusion in epidural analgesia. However, it is known for a higher risk of cardiotoxicity compared to other local anesthetics, especially when inadvertently injected intravascularly or in high doses, necessitating careful dosing and monitoring. Pharmacokinetically, bupivacaine is absorbed from the injection site at a rate dependent on the vascularity of the tissue. It is extensively bound to plasma proteins, primarily alpha-1 acid glycoprotein, which affects its distribution and duration. Bupivacaine is metabolized hepatically via cytochrome P450 enzymes to inactive metabolites that are excreted mainly in urine. The elimination half-life ranges between 2.7 to 3.5 hours. Adverse effects associated with bupivacaine include central nervous system symptoms such as dizziness, tinnitus, or seizures in cases of systemic toxicity. Cardiovascular effects can be severe, including arrhythmias and cardiac depression, which have led to the development of lipid emulsion therapy as an effective treatment for bupivacaine-induced cardiotoxicity. Bupivacaine’s clinical use requires appropriate technique to avoid complications, including aspiration before injection to prevent intravascular administration and dose adjustment in patients with hepatic impairment or other risk factors. Its efficacy and duration have made it a cornerstone in regional anesthesia practice. In summary, bupivacaine is a long-acting amide local anesthetic widely used for regional anesthesia and analgesia. Its potent anesthetic effect, combined with prolonged duration, supports its use in major surgical and obstetric procedures, while its known toxicity profile necessitates careful clinical management. References 1979. Melanin Capacity to Accumulate Drugs in the Internal Ear: A Study on Lidocaine, Bupivacaine and Chlorpromazine. Acta Oto-Laryngologica, 88(1-2). DOI: 10.3109/00016487909137141 1991. Electrophysiologic effects of bupivacaine in the isolated rabbit heart. Anesthesia and Analgesia, 72(1). DOI: 1984378 2000. Bupivacaine Release from Biopolymeric Depots for the Alleviation of Postoperative Pain. Biomaterials Engineering and Devices: Human Applications. DOI: 10.1007/978-1-59259-196-1_19 |
| Market Analysis Reports |