Online Database of Chemicals from Around the World
| Luminescence Technology Corp. | Taiwan | |||
|---|---|---|---|---|
 | www.lumtec.com.tw | |||
 | +886 (2) 2697-5600 | |||
 | +886 (2) 2697-5601 | |||
 | sales@lumtec.com.tw | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | (Quinolin-8-olato)lithium |
| Synonyms | Liq |
| Molecular Structure | 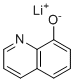 |
| Molecular Formula | C9H6LiNO |
| Molecular Weight | 151.09 |
| CAS Registry Number | 25387-93-3 |
| EC Number | 246-932-3 |
| SMILES | [Li+].C1=CC2=C(C(=C1)[O-])N=CC=C2 |
| Melting point | 363-368 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
(Quinolin-8-olato)lithium, commonly referred to as lithium 8-hydroxyquinolate, is an organolithium compound derived from quinolin-8-ol, also known as 8-hydroxyquinoline. This compound has garnered significant attention in various chemical and industrial fields due to its unique structural features and functional versatility. The molecule consists of a quinoline ring system, with a hydroxyl group at the 8-position coordinating to a lithium cation, forming a stable chelate structure. The ability of the ligand to act as a bidentate species contributes to the compound's stability and reactivity. The discovery of (quinolin-8-olato)lithium can be traced back to studies involving the coordination chemistry of 8-hydroxyquinoline and its derivatives. During the mid-20th century, researchers explored the complex-forming ability of 8-hydroxyquinoline with various metals, leading to the synthesis of corresponding metal chelates, including those of lithium. The ability of 8-hydroxyquinoline to chelate metal ions arises from the presence of both a nitrogen atom and a phenolic oxygen atom, enabling stable coordination through a five-membered ring. One of the prominent applications of (quinolin-8-olato)lithium is in organic synthesis, particularly in facilitating metallation reactions. The lithium center enhances the nucleophilicity of the aromatic ring, making it a useful intermediate for selective lithiation processes. These reactions are valuable in producing functionalized quinoline derivatives, which are important in the pharmaceutical and material industries. The reactivity of (quinolin-8-olato)lithium allows for subsequent transformations, including coupling reactions, halogenation, and acylation, thereby expanding the range of synthetic possibilities. In pharmaceutical chemistry, quinoline derivatives have long been recognized for their broad biological activity, including antimicrobial, antimalarial, and anticancer properties. By employing (quinolin-8-olato)lithium in the synthesis of these derivatives, researchers can introduce functional groups at precise positions on the quinoline ring, optimizing the biological activity of the resulting molecules. The compound’s utility in lithiation and functionalization processes makes it a valuable tool for medicinal chemists developing new drug candidates. Another area where (quinolin-8-olato)lithium finds application is in materials science. Quinoline-based compounds are known for their optical and electronic properties, making them suitable for applications in organic light-emitting diodes (OLEDs) and fluorescent materials. The lithium chelate of 8-hydroxyquinoline exhibits luminescent properties, which can be harnessed in the development of light-emitting materials and sensors. The presence of lithium, a light alkali metal, also ensures that the resulting materials maintain a low molecular weight and high thermal stability. Moreover, (quinolin-8-olato)lithium has been explored in the field of catalysis. Organolithium reagents are known for their role in initiating polymerization reactions, particularly in the synthesis of polymers with well-defined architectures. The chelating nature of the quinoline ligand can modify the reactivity of the lithium center, allowing for controlled polymerization processes. This property is useful in producing specialized polymers with tailored properties for industrial applications. The synthesis of (quinolin-8-olato)lithium typically involves the deprotonation of 8-hydroxyquinoline using lithium bases such as n-butyllithium or lithium hydride. The reaction proceeds under mild conditions and results in the formation of the lithium chelate, which can be isolated and purified for further use. Advances in synthetic methodologies have improved the efficiency and scalability of this process, making the compound readily available for laboratory and industrial applications. Research into (quinolin-8-olato)lithium continues to expand as scientists explore new applications and optimize existing processes. Its role in organic synthesis, pharmaceutical development, materials science, and catalysis underscores its versatility and importance. The compound’s stability, reactivity, and functional adaptability ensure its continued relevance in both academic research and industrial applications. References 2020. Organic light emitters exhibiting very fast reverse intersystem crossing. Nature Photonics, 14(10). DOI: 10.1038/s41566-020-0667-0 2020. Promising applications of aggregation-induced emission luminogens in organic optoelectronic devices. PhotoniX, 1(1). DOI: 10.1186/s43074-020-00012-y 2012. Carbazolyl Benzo[1,2-b:4,5-b′]difuran: An Ambipolar Host Material for Full-Color Organic Light-Emitting Diodes. Chemistry - An Asian Journal, 7(6). DOI: 10.1002/asia.201200062 |
| Market Analysis Reports |