Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | 6-Bromoisoquinoline |
| Molecular Structure | 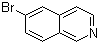 |
| Molecular Formula | C9H6BrN |
| Molecular Weight | 208.05 |
| CAS Registry Number | 34784-05-9 |
| EC Number | 662-848-3 |
| SMILES | C1=CC2=C(C=CN=C2)C=C1Br |
| Melting point | 45-46 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P305+P351+P338-P330-P337+P317-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
6-Bromoisoquinoline is an important chemical compound in the field of organic chemistry and medicinal chemistry due to its unique structure and diverse applications. This compound features an isoquinoline ring system—a fused bicyclic structure containing a benzene ring and a pyridine ring—with a bromo substituent positioned at the 6th carbon atom. The discovery of 6-bromoisoquinoline can be linked to the broader study of halogenated isoquinolines, which have been investigated for their potential biological activities and chemical reactivity. Isoquinolines themselves are a class of heterocyclic compounds that have been widely studied for their pharmacological properties and utility in organic synthesis. The introduction of the bromine atom at the 6-position of the isoquinoline ring imparts unique electronic and steric effects, which can significantly influence the compound's reactivity and interactions with biological targets. This halogenation introduces additional opportunities for chemical modification and functionalization, making 6-bromoisoquinoline a valuable intermediate in various chemical processes. One of the primary applications of 6-bromoisoquinoline is in medicinal chemistry, where it serves as a key building block for the synthesis of bioactive compounds. The presence of the bromine atom enhances the electrophilic character of the ring, facilitating nucleophilic substitution reactions that are useful in creating diverse chemical derivatives. These derivatives can be screened for potential therapeutic properties, including antimicrobial, anticancer, and anti-inflammatory activities. For instance, compounds derived from 6-bromoisoquinoline have shown promise in inhibiting certain enzymes and receptors, contributing to the development of novel drugs. In addition to its role in drug discovery, 6-bromoisoquinoline is employed in organic synthesis as a reagent or intermediate for creating complex molecules. Its unique structure allows it to participate in various chemical reactions, including cross-coupling reactions and functional group transformations. This versatility is particularly advantageous in the synthesis of complex organic compounds used in materials science and chemical research. The synthesis of 6-bromoisoquinoline typically involves the bromination of isoquinoline using brominating agents under controlled conditions. The reaction conditions, such as temperature and solvent choice, are crucial for achieving high yields and selectivity. The resulting compound can then be used as a precursor for further chemical modifications or as an intermediate in the synthesis of more complex molecules. In summary, 6-bromoisoquinoline is a significant compound in both medicinal chemistry and organic synthesis. Its unique structural features, combined with the presence of the bromine atom, provide valuable opportunities for the development of new drugs and the synthesis of complex organic molecules. The ongoing exploration of its applications and derivatives continues to advance both pharmaceutical research and chemical synthesis. References 2021. Functionalization of Isoquinolines. Synlett, 32(10). DOI: 10.1055/s-0040-1706543 |
| Market Analysis Reports |