Online Database of Chemicals from Around the World
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2016 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | 5-Bromoisoquinoline |
| Molecular Structure | 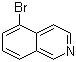 |
| Molecular Formula | C9H6BrN |
| Molecular Weight | 208.05 |
| CAS Registry Number | 34784-04-8 |
| EC Number | 626-788-1 |
| SMILES | C1=CC2=C(C=CN=C2)C(=C1)Br |
| Density | 1.6±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 83-87 °C (Expl.) |
| Index of Refraction | 1.674, Calc.* |
| Boiling Point | 312.3±15.0 °C (760 mmHg), Calc.*, 95-97 °C (0.1 mmHg) (Expl.) |
| Flash Point | 142.7±20.4 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
5-Bromoisoquinoline is an organic compound with the molecular formula C9H6BrN. It consists of an isoquinoline ring structure, a nitrogen-containing heterocycle, with a bromine atom attached at the 5-position on the benzene ring. This chemical belongs to the broader class of halogenated isoquinolines, which are known for their diverse biological activities and their utility in synthetic chemistry. The discovery of 5-bromoisoquinoline can be attributed to the ongoing exploration of halogenated heterocycles, which are valuable due to their ability to influence reactivity and interaction with biological targets. The synthesis of 5-bromoisoquinoline typically involves bromination of isoquinoline, a well-known reaction in organic chemistry that introduces a bromine atom at the desired position on the aromatic ring. This reaction is often carried out using bromine or bromine-containing reagents in the presence of a solvent such as acetic acid, which facilitates the electrophilic aromatic substitution. The position of the bromine on the aromatic ring can be controlled to a certain extent by optimizing reaction conditions, making this a versatile synthetic route for obtaining 5-bromoisoquinoline. The applications of 5-bromoisoquinoline are widespread across different areas of chemical research and industry. One of the primary applications is in the field of medicinal chemistry, where it serves as an important intermediate for the synthesis of bioactive compounds. Halogenation of isoquinoline derivatives, like the introduction of the bromine atom at the 5-position, often enhances the biological activity of the resulting molecules. Studies have shown that 5-bromoisoquinoline and its derivatives exhibit antimicrobial, anti-inflammatory, and anticancer properties, making them potential candidates for drug development. Specifically, compounds derived from 5-bromoisoquinoline have shown promise in inhibiting certain kinases and enzymes, which are critical targets in cancer therapy. In addition to its biomedical applications, 5-bromoisoquinoline is valuable in the development of organic materials, such as organic semiconductors and light-emitting diodes (LEDs). The presence of a bromine atom in the isoquinoline structure can modify the electronic properties of the compound, making it suitable for use in organic electronics. The compound can be incorporated into polymeric materials or used as a building block in the synthesis of novel organic electronic devices, where its ability to participate in π-π stacking interactions contributes to improved charge transport and device performance. Furthermore, 5-bromoisoquinoline plays a significant role in synthetic organic chemistry as a versatile building block. Its structure makes it an excellent candidate for further functionalization, enabling the creation of a wide range of complex molecules with various substituents. The bromine atom serves as a useful site for nucleophilic substitution reactions, which can be used to introduce additional functional groups. This makes 5-bromoisoquinoline a key intermediate in the synthesis of various compounds, including pharmaceuticals, agrochemicals, and advanced materials. In conclusion, 5-bromoisoquinoline is a crucial compound in both medicinal chemistry and material science. Its ability to serve as a synthetic intermediate and its promising biological and electronic properties make it an important target in research and development. The ongoing exploration of its derivatives continues to offer potential in drug discovery and the creation of new functional materials. References 2021. Functionalization of Isoquinolines. Synlett, 32(10). DOI: 10.1055/s-0040-1706543 |
| Market Analysis Reports |