Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Frontier Specialty Chemicals | USA | |||
|---|---|---|---|---|
 | www.frontierspecialtychemicals.com | |||
 | +1 (435) 753-1901 | |||
 | +1 (435) 753-6731 | |||
 | sales@frontierspecialtychemicals.com | |||
| Chemical manufacturer since 1975 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Isoquinoline compound |
|---|---|
| Name | 7-Bromoisoquinoline |
| Molecular Structure | 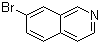 |
| Molecular Formula | C9H6BrN |
| Molecular Weight | 208.05 |
| CAS Registry Number | 58794-09-5 |
| EC Number | 676-321-0 |
| SMILES | C1=CC(=CC2=C1C=CN=C2)Br |
| Density | 1.6±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.674, Calc.* |
| Melting point | 67-72 °C (Expl.) |
| Boiling Point | 312.3±15.0 °C (760 mmHg), Calc.* |
| Flash Point | 142.7±20.4 °C, Calc.*, 113 °C (Expl.) |
| Water solubility | insoluble |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
7-Bromoisoquinoline is an organic compound that consists of an isoquinoline ring system, which is a bicyclic structure composed of a benzene ring fused to a pyridine ring. The compound is substituted at the 7-position of the isoquinoline ring with a bromine atom, which significantly alters its chemical reactivity and makes it useful in various synthetic applications. The discovery and synthesis of 7-bromoisoquinoline can be traced back to early studies in heterocyclic chemistry, where it was recognized as an important derivative of isoquinoline. Isoquinoline and its derivatives have long been of interest due to their biological activity and use as intermediates in the synthesis of pharmaceuticals and agrochemicals. 7-Bromoisoquinoline is primarily used as an intermediate in organic synthesis. The presence of the bromine atom at the 7-position of the isoquinoline ring makes it highly reactive in nucleophilic aromatic substitution reactions. This reactivity allows it to be used as a starting material for the synthesis of more complex molecules, particularly in the pharmaceutical and agrochemical industries. One of the key applications of 7-bromoisoquinoline is in the synthesis of biologically active compounds. Isoquinoline derivatives have been widely studied for their medicinal properties, and the 7-bromo substitution provides a versatile platform for modifying the isoquinoline structure to create compounds with specific biological activities. For example, it has been employed in the synthesis of potential anti-cancer and anti-inflammatory agents. The bromine atom facilitates the introduction of various functional groups through substitution reactions, enabling the development of new pharmaceutical molecules. In addition to its role in drug synthesis, 7-bromoisoquinoline is also used in the production of dyes and pigments. Isoquinoline derivatives have been shown to exhibit interesting fluorescence properties, making them useful in the development of organic fluorescent materials. These materials have applications in organic light-emitting diodes (OLEDs) and other optoelectronic devices, where their ability to emit light in response to electrical stimuli is valuable. 7-Bromoisoquinoline also finds use in the development of new chemical reactions. It serves as a model compound for studying nucleophilic aromatic substitution mechanisms, and it is used in research to explore the reactivity of halogenated heterocyclic compounds. The reactivity of the bromo group at the 7-position makes it a useful reagent in the synthesis of other isoquinoline derivatives, allowing for the introduction of diverse substituents that can alter the compound's properties. Furthermore, 7-bromoisoquinoline has been employed in the synthesis of materials used in catalysis and sensors. Its ability to coordinate with metal ions allows it to serve as a ligand in the preparation of metal complexes, which are important in catalytic processes. These complexes are used in a wide range of industrial applications, including in the production of fine chemicals and in environmental remediation. In summary, 7-bromoisoquinoline is an important intermediate in organic synthesis with applications in drug development, materials science, and catalysis. Its reactivity, particularly in nucleophilic aromatic substitution, makes it a valuable building block for the synthesis of complex organic molecules. The compound's well-established uses in the synthesis of biologically active compounds, dyes, pigments, and metal complexes underscore its versatility and importance in both academic and industrial research. References 2011. Efficient Carbonylation of Aryl and Heteroaryl Bromides under Atmospheric Pressure of CO. Synlett, 2011(14). DOI: 10.1055/s-0030-1261193 2013. Enantioselective Reactions of N‐Acyliminium Ions Using Chiral Organocatalysts. Chemistry - An Asian Journal, 8(11). DOI: 10.1002/asia.201300814 |
| Market Analysis Reports |