Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | 8-Bromoisoquinoline |
| Molecular Structure | 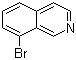 |
| Molecular Formula | C9H6BrN |
| Molecular Weight | 208.06 |
| CAS Registry Number | 63927-22-0 |
| EC Number | 689-822-4 |
| SMILES | C1=CC2=C(C=NC=C2)C(=C1)Br |
| Melting point | 80 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
8-Bromoisoquinoline is a chemical compound with a distinctive structure and notable applications in synthetic chemistry and medicinal research. Its discovery and development have contributed to advancements in the field of organic chemistry, particularly in the synthesis of complex molecules and the study of biologically active substances. The discovery of 8-Bromoisoquinoline can be traced back to the early 20th century, during a period of intensive research into aromatic heterocycles. The compound is a brominated derivative of isoquinoline, a nitrogen-containing aromatic heterocycle. The introduction of the bromine atom at the 8-position of the isoquinoline ring system provides the compound with unique chemical reactivity and properties. This substitution pattern was first identified and characterized through the synthesis of isoquinoline derivatives and subsequent bromination reactions. In terms of applications, 8-Bromoisoquinoline is widely used in synthetic organic chemistry. Its reactivity allows it to serve as a valuable intermediate in the synthesis of more complex molecules. For example, the bromine atom in 8-Bromoisoquinoline can participate in various coupling reactions, such as Suzuki and Sonogashira couplings, which are essential for constructing carbon-carbon bonds in organic synthesis. These reactions are instrumental in the creation of pharmaceuticals, agrochemicals, and advanced materials. Moreover, 8-Bromoisoquinoline plays a significant role in medicinal chemistry. Its structural similarity to isoquinoline, which is a core structure in numerous biologically active compounds, makes it a useful building block for the development of novel drugs. Researchers have explored its potential as a precursor in the synthesis of isoquinoline-based pharmaceuticals, including those with anticancer, antimicrobial, and anti-inflammatory properties. The bromine substitution can also enhance the compound's interaction with biological targets, potentially improving the efficacy and selectivity of therapeutic agents. The compound's role extends to the study of biological systems and chemical biology. 8-Bromoisoquinoline is used as a probe in investigations of enzyme mechanisms and cellular processes. Its ability to selectively interact with certain proteins and enzymes allows scientists to explore biochemical pathways and develop new methods for probing cellular functions. The synthesis of 8-Bromoisoquinoline typically involves the bromination of isoquinoline. This process is carried out using bromine or brominating agents under controlled conditions to ensure selective substitution at the 8-position of the isoquinoline ring. Characterization of the product is performed using techniques such as nuclear magnetic resonance (NMR) spectroscopy, mass spectrometry, and infrared spectroscopy to confirm the structure and purity of the compound. In summary, 8-Bromoisoquinoline is a versatile compound with important applications in synthetic chemistry and medicinal research. Its discovery and development have provided valuable tools for constructing complex molecules and studying biological systems, contributing to advancements in chemistry and drug development. References 2013. Enantioselective Reactions of N‐Acyliminium Ions Using Chiral Organocatalysts. Chemistry - An Asian Journal, 8(11). DOI: 10.1002/asia.201300814 2018. For Thioetherification Reactions. Science of Synthesis. https://science-of-synthesis.thieme.com 2020. Synthesis of A-1331852. Synfacts, 16(12). DOI: 10.1055/s-0040-1706390 2023. Cycloalkanes Containing an Heteroatom (Heterocyclic Compounds). Alicyclic Chemistry. DOI: 10.1007/978-3-031-36068-8_9 |
| Market Analysis Reports |