Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hangzhou Luna Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lunachem.com | |||
 | +86 (571) 8577-5970 8577-5800 8577-5660 | |||
 | +86 (571) 8577-5960 | |||
 | tan@lunachem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Jiangsu Nhwa Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nhwa-group.com | |||
 | +86 (516) 8766-1170 | |||
 | +86 (516) 8776-9782 | |||
 | nhwasale@nhwa-group.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| D.K. Pharma Chem Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.dkpharmachem.com | |||
 | +91 (251) 269-0111 | |||
 | +91 (251) 269-0366 | |||
 | marketing@dkpharmachem.com | |||
| Chemical manufacturer since 1980 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Wuhan Yuancheng Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuanchengtechnology.com | |||
 | +86 17282536078 | |||
 | +86 17282536078 | |||
 | 3042184429@qq.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17282536078 | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| Paragos e. K. | Germany | |||
|---|---|---|---|---|
 | www.paragos.com | |||
 | +49 (2330) 807-9751 | |||
 | +49 (2330) 807-9752 | |||
 | sales@paragos.de | |||
| Chemical manufacturer since 2001 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Hubei Teyer Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hubeiteyer.com | |||
 | +86 (728) 533-2622 | |||
 | info@hubeiteyer.com | |||
| Chemical manufacturer since 2002 | ||||
| Zhejiang Excel Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.excelpharma.com | |||
 | +86 (576) 8416-0008 | |||
 | +86 (576) 8416-0000 | |||
 | sales@excelpharma.com | |||
| Chemical manufacturer since 2000 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Circulatory system medication >> Regulating blood lipids |
|---|---|
| Name | Fenofibrate |
| Synonyms | 2-[4-(4-Chlorobenzoyl)phenoxy]-2-methylpropanoic acid isopropyl ester; Proctofene; Sedufen |
| Molecular Structure | 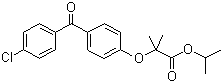 |
| Molecular Formula | C20H21ClO4 |
| Molecular Weight | 360.84 |
| CAS Registry Number | 49562-28-9 |
| EC Number | 256-376-3 |
| SMILES | CC(C)OC(=O)C(C)(C)OC1=CC=C(C=C1)C(=O)C2=CC=C(C=C2)Cl |
| Solubility | 100 mM (DMSO), 100 mM (ethanol) (Expl.) |
|---|---|
| Density | 1.2±0.1 g/cm3, Calc.* |
| Melting Point | 81 °C (Expl.) |
| Index of Refraction | 1.547, Calc.* |
| Boiling Point | 469.8±35.0 °C (760 mmHg), Calc.* |
| Flash Point | 165.4±24.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Warning Details GHS08 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H373-H413 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P260-P273-P319-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Fenofibrate is a medication primarily used to reduce cholesterol levels in the blood. It belongs to a class of drugs known as fibrates, which are commonly prescribed to manage dyslipidemia, a condition characterized by abnormal levels of lipids, such as cholesterol and triglycerides, in the blood. Fenofibrate is used in the treatment of hypertriglyceridemia and mixed dyslipidemia, often as an adjunct to dietary changes and exercise. Fenofibrate works by activating peroxisome proliferator-activated receptor alpha (PPAR-α), a nuclear receptor that plays a key role in the regulation of lipid metabolism. Activation of PPAR-α leads to an increase in the expression of genes involved in the breakdown of fatty acids and the reduction of triglyceride synthesis in the liver. As a result, fenofibrate helps lower triglyceride levels and increase high-density lipoprotein (HDL) cholesterol, often referred to as "good" cholesterol. In addition, fenofibrate may modestly reduce low-density lipoprotein (LDL) cholesterol, known as "bad" cholesterol. The primary clinical benefit of fenofibrate is its ability to lower triglyceride levels, which is particularly important for patients at risk of cardiovascular disease, as elevated triglycerides are a significant risk factor. The medication is also used to increase HDL cholesterol, which has protective effects on the cardiovascular system by helping to remove excess cholesterol from the bloodstream and prevent the buildup of plaques in the arteries. Fenofibrate is typically administered orally in the form of tablets or capsules. It is often prescribed when other measures, such as diet and exercise, have been insufficient to manage lipid levels. The medication is generally well-tolerated, but like all drugs, it can cause side effects in some patients. Common side effects include gastrointestinal issues such as nausea, diarrhea, and abdominal pain. More serious side effects, although rare, can include liver dysfunction, muscle pain (myopathy), and kidney problems, especially in patients with pre-existing kidney disease. One important aspect of fenofibrate therapy is that it is often used in combination with other lipid-lowering agents, such as statins, for more effective management of dyslipidemia. However, the combination of fenofibrate and statins should be carefully monitored due to the potential increased risk of muscle-related side effects, particularly in older adults or those with kidney impairment. In addition to its role in lipid management, fenofibrate has been studied for its potential to reduce the risk of atherosclerosis and cardiovascular events. Some studies have suggested that fenofibrate may help reduce inflammation and improve endothelial function, which could contribute to its cardiovascular protective effects. However, its role in primary prevention of cardiovascular disease remains less well-established compared to other therapies, such as statins. In summary, fenofibrate is a widely used medication for managing hypertriglyceridemia and mixed dyslipidemia. It works by activating PPAR-α to reduce triglyceride levels and increase HDL cholesterol, thus improving lipid profiles and reducing the risk of cardiovascular disease. While it is generally well-tolerated, it can cause side effects, and its use should be closely monitored, especially in combination with other lipid-lowering drugs like statins. References 1998. Long-Term Efficacy and Safety of Fenofibrate and a Statin in the Treatment of Combined Hyperlipidemia. The American Journal of Cardiology, 81(4). DOI: 10.1016/s0002-9149(98)00040-x 1998. Clinical Pharmacokinetics of Fibric Acid Derivatives (Fibrates). Clinical Pharmacokinetics, 34(2). DOI: 10.2165/00003088-199834020-00003 1984. Fenofibrate therapy of hypertriglyceridaemias. Differential effects on LDL cholesterol level in Type IV and in Type IIb primary hyperlipoproteinaemia. European Journal of Clinical Pharmacology, 27(5). DOI: 10.1007/bf00541935 |
| Market Analysis Reports |