Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Ketones |
|---|---|
| Name | 10-Phenyl-9(10H)-acridinone |
| Synonyms | 10-Phenylacridone; N-Phenylacridone; NSC 27927; Phenylacridone |
| Molecular Structure | 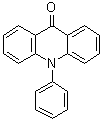 |
| Molecular Formula | C19H13NO |
| Molecular Weight | 271.31 |
| CAS Registry Number | 5472-23-1 |
| EC Number | 878-853-0 |
| SMILES | C1=CC=C(C=C1)N2C3=CC=CC=C3C(=O)C4=CC=CC=C42 |
| Solubility | Insoluble (1.5E-3 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.245±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 278 °C** (Expl.) |
| Index of Refraction | 1.676, Calc.* |
| Boiling Point | 428.1±15.0 °C (760 mmHg), Calc.* |
| Flash Point | 169.6±9.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2017 ACD/Labs) |
| ** | Storoniak, P. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
10-Phenyl-9(10H)-acridinone is an organic compound that belongs to the class of acridone derivatives. The chemical formula of 10-phenyl-9(10H)-acridinone is C15H11NO, and it consists of a phenyl group attached to the 10 position of the acridone structure. Acridone itself is a heterocyclic compound featuring a nitrogen-containing ring system, and the substitution of a phenyl group at the 10-position enhances its chemical properties and potential applications. The discovery of 10-phenyl-9(10H)-acridinone traces back to the study of acridone derivatives, which has been a subject of interest due to their wide range of biological and chemical activities. Acridones were first investigated in the early 20th century, and over time, various substituted acridones, including 10-phenyl-9(10H)-acridinone, were synthesized to explore their potential applications in medicinal chemistry and materials science. The introduction of a phenyl group into the acridone framework further increases the compound's stability and introduces specific reactivity patterns that make it useful in diverse synthetic and catalytic processes. 10-Phenyl-9(10H)-acridinone has found significant application in the field of organic electronics, particularly in the development of organic light-emitting diodes (OLEDs). OLEDs are a type of display technology that is widely used in various consumer electronics, including smartphones, televisions, and digital displays. Acridone derivatives like 10-phenyl-9(10H)-acridinone are valued for their ability to emit light efficiently when subjected to an electric current, making them useful in the creation of high-performance OLED materials. The phenyl substitution on the acridone structure helps to fine-tune the optical properties of the compound, leading to improved light emission and color purity in OLED devices. Another important application of 10-phenyl-9(10H)-acridinone is in the field of medicinal chemistry, where it has shown promise as an anticancer agent. Acridone derivatives have long been studied for their ability to inhibit the growth of cancer cells through various mechanisms, such as intercalating with DNA and interfering with cellular processes. The phenyl substitution in 10-phenyl-9(10H)-acridinone can modulate its biological activity, making it a potential lead compound for the development of novel anticancer drugs. Studies have shown that compounds like 10-phenyl-9(10H)-acridinone may be able to selectively target cancer cells, leading to reduced toxicity compared to conventional chemotherapy agents. Additionally, 10-phenyl-9(10H)-acridinone has been investigated for its fluorescence properties, which make it useful in the development of chemical sensors and imaging agents. Its ability to interact with biological molecules and emit fluorescence upon excitation enables its use in biological and environmental sensing applications. References 2012. Fluorescence Probe of 10-Phenyl-acridone-2-sulfonyl Chloride and Its Application for Determination of Free Aliphatic Amines in Environmental Samples by HPLC with Fluorescence Detection and APCI-MS. Chromatographia, 75(3). DOI: 10.1007/s10337-012-2298-1 2003. Melting, volatilisation and crystal lattice enthalpies of acridin-9(10H)-ones. Journal of Thermal Analysis and Calorimetry, 74(1). DOI: 10.1023/b:jtan.0000005179.91819.6d 1965. Research in the field of heterocyclic chemistry. XXXVIII. Mechanism of the acridone synthesis based on diphenylamine-2-carboxylic acids. Chemistry of Heterocyclic Compounds, 1(2). DOI: 10.1007/bf01046679 |
| Market Analysis Reports |