Online Database of Chemicals from Around the World
| Glentham Life Sciences | UK | |||
|---|---|---|---|---|
 | www.glenthamls.com | |||
 | +44 (2033) 978-798 | |||
 | +44 (2033) 978-909 | |||
 | info@glenthamls.com | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Dalton Pharma Services | Canada | |||
|---|---|---|---|---|
 | www.dalton.com | |||
 | +1 (416) 661-2102 | |||
 | +1 (416) 661-2108 | |||
 | chemist@dalton.com | |||
| Chemical manufacturer | ||||
| Jiangxi Biochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biochemjx.com | |||
 | +86 (791) 662-9460 | |||
 | +86 (791) 664-0053 | |||
 | lfl@biochemjx.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Hormone and endocrine-regulating drugs |
|---|---|
| Name | 11b-Hydroxyandrost-4-ene-3,17-dione |
| Synonyms | 11-Hydroxy-10,13-dimethyl-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthrene-3,17-dione |
| Molecular Structure | 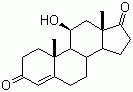 |
| Molecular Formula | C19H26O3 |
| Molecular Weight | 302.41 |
| CAS Registry Number | 564-33-0 |
| SMILES | C[C@]12CCC(=O)C=C1CC[C@@H]3[C@@H]2[C@@H](C[C@]4([C@H]3CCC4=O)C)O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.569, Calc.* |
| Boiling Point | 475.3±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 255.4±25.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P351-P302+P352 Details |
| SDS | Available |
|
11β-Hydroxyandrost-4-ene-3,17-dione (11β-OHA), a hydroxylated derivative of androst-4-ene-3,17-dione, is a key intermediate in the biosynthesis of adrenal cortical steroids. It was first discovered in the early 20th century through studies of steroid metabolism. This breakthrough came from the work of Butenandt and Ruzicka, who played a major role in the elucidation of steroid structure. In the 1930s, studies of adrenal cortical hormones revealed numerous intermediates. 11β-OHA was isolated during these explorations, highlighting its role in steroid biosynthesis. Its identification provided insights into the pathways for the production of cortisol and other corticosteroids, emphasizing the importance of hydroxylation at the C11 position in steroid metabolism. 11β-OHA plays a key role in the synthesis of anti-inflammatory corticosteroids such as prednisone and dexamethasone. These drugs are widely used for their potent anti-inflammatory and immunosuppressive properties to treat diseases such as asthma, rheumatoid arthritis, and certain cancers. In biochemical and pharmacological studies, 11β-OHA is a key compound in the study of the steroidogenic pathway. Its role in understanding the function of enzymes such as 11β-hydroxylase is valuable in developing treatments for adrenal disorders. The relevance of 11β-OHA extends to diagnostics, particularly in the assessment of adrenal function. Measuring its levels helps diagnose diseases such as Cushing's syndrome and Addison's disease, which result in disruptions in cortisol biosynthesis. In biotechnology, 11β-OHA is used in the microbial conversion of steroids. Certain microorganisms can convert it into other valuable steroids, enhancing its importance in industrial applications. References 2023. Current Trends and Perspectives in Microbial Bioconversions of Steroids. Methods in molecular biology (Clifton, N.J.), 2704. DOI: 10.1007/978-1-0716-3385-4_1 2017. Obtaining of 11α-Hydroxyandrost-4-ene-3,17-dione from Natural Sterols. Methods in molecular biology (Clifton, N.J.), 1645. DOI: 10.1007/978-1-4939-7183-1_18 2019. Microbial transformation of cholesterol: reactions and practical aspects—an update. World Journal of Microbiology and Biotechnology, 35(8). DOI: 10.1007/s11274-019-2708-8 |
| Market Analysis Reports |