Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Chemical reagent >> Organic reagent >> Tricyclic compound |
|---|---|
| Name | 9-Methylacridine |
| Synonyms | NSC 1234 |
| Molecular Structure | 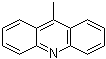 |
| Molecular Formula | C14H11N |
| Molecular Weight | 193.24 |
| CAS Registry Number | 611-64-3 |
| EC Number | 210-272-4 |
| SMILES | CC1=C2C=CC=CC2=NC3=CC=CC=C13 |
| Solubility | Practically insoluble (0.039 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.155±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 129-130 °C** |
| Index of Refraction | 1.703, Calc.* |
| Boiling Point | 361.9±11.0 °C (760 mmHg), Calc.* |
| Flash Point | 160.0±11.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| ** | Tsvelikhovsky, Dmitry |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
9-Methylacridine is a chemical compound belonging to the class of acridines, which are aromatic compounds containing a nitrogen atom within a three-ring structure. It consists of a methyl group attached to the ninth position of the acridine ring system. This modification of the acridine structure has significant implications for the chemical properties and reactivity of the molecule. 9-Methylacridine was first synthesized in the mid-20th century during the exploration of substituted acridines, a class of compounds known for their diverse chemical reactivity and biological activities. Since its discovery, 9-methylacridine has been studied for its applications in organic synthesis, material science, and medicinal chemistry. One of the main applications of 9-methylacridine lies in its use as an intermediate in organic synthesis. Due to its unique chemical structure, it is a valuable building block for the synthesis of more complex compounds. 9-Methylacridine can participate in a variety of reactions, including nucleophilic substitution, electrophilic aromatic substitution, and other types of functional group transformations. These reactions enable the synthesis of other acridine derivatives, which are used in the production of dyes, agrochemicals, and pharmaceuticals. Its ability to form derivatives with various functional groups makes it a versatile compound in synthetic chemistry. In addition to its use in organic synthesis, 9-methylacridine has found applications in the field of medicinal chemistry. Acridine derivatives, including 9-methylacridine, are known for their antimicrobial, anticancer, and antimalarial properties. The structure of 9-methylacridine allows it to interact with biological systems in ways that can inhibit the growth of pathogens or cancer cells. Researchers have explored the potential of 9-methylacridine and its derivatives as therapeutic agents, particularly in the development of drugs aimed at treating cancer and infectious diseases. Acridine-based compounds can intercalate into DNA, affecting the replication process, which makes them useful as chemotherapeutic agents. 9-Methylacridine has also been studied for its role in the development of optoelectronic materials. The presence of a nitrogen atom in the acridine ring and the methyl group at the ninth position can influence the electronic properties of the molecule, making it a candidate for use in light-emitting devices, organic semiconductors, and other electronic applications. The compound’s ability to absorb and emit light at specific wavelengths makes it useful in the design of organic light-emitting diodes (OLEDs) and organic solar cells. These materials are of particular interest due to their potential for use in flexible electronics and energy-efficient devices. Moreover, 9-methylacridine has been investigated for its potential use in fluorescence-based sensors and detection systems. Its fluorescence properties, when exposed to certain wavelengths of light, can be harnessed for sensing applications, such as the detection of specific metal ions or other environmental pollutants. By modifying the structure of 9-methylacridine, researchers have been able to design sensors that can selectively bind to target molecules, providing a useful tool for environmental monitoring and diagnostic applications. In conclusion, 9-methylacridine is a valuable chemical compound with a wide range of applications in organic synthesis, medicinal chemistry, material science, and sensor technology. Its unique structure allows it to participate in various chemical reactions and interact with biological systems, making it an important building block for the development of new therapeutic agents and functional materials. As research continues to explore its potential, 9-methylacridine is likely to remain an important compound in both academic and industrial settings. References 2019. Recent Advances in the Synthesis of Acridines and Phenazines. *Synlett*, 31(01). DOI: 10.1055/s-0039-1690708 2023. Cytotoxicity of a new spiro-acridine derivative: modulation of cellular antioxidant state and induction of cell cycle arrest and apoptosis in HCT-116 colorectal carcinoma. *Naunyn-Schmiedeberg's Archives of Pharmacology*. DOI: 10.1007/s00210-023-02686-0 1954. Condensation of benzo derivatives of 9-methylacridine. *Bulletin of the Academy of Sciences of the USSR, Division of chemical science*. DOI: 10.1007/bf01172699 |
| Market Analysis Reports |