Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shenzhen BST Science & Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bstcom.cn | |||
 | +86 (755) 2553-7800 | |||
 | +86 (755) 2553-9493 | |||
 | good618@126.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Antiparasitic drug >> Anthelmintic medications |
|---|---|
| Name | Ansamitocin P 3 |
| Synonyms | Ansamitosin P 3; Antibiotic C 15003P3; Maytansinol isobutyrate; NSC 292222; 2'-De(acetylmethylamino)-2'-methylmaytansine |
| Molecular Structure | 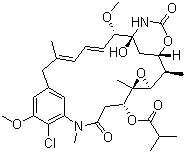 |
| Molecular Formula | C32H43ClN2O9 |
| Molecular Weight | 635.14 |
| CAS Registry Number | 66584-72-3 |
| EC Number | 680-663-6 |
| SMILES | C[C@@H]1[C@@H]2C[C@]([C@@H](/C=C/C=C(/CC3=CC(=C(C(=C3)OC)Cl)N(C(=O)C[C@@H]([C@]4([C@H]1O4)C)OC(=O)C(C)C)C)C)OC)(NC(=O)O2)O |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.583, Calc.* |
| Boiling Point | 833.1±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 457.7±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H332-H302-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Ansamitocin P 3 is a member of the ansamycin family of compounds originally isolated from Actinosynnema pretiosum, a soil bacterium, in the 1970s. Its discovery was driven by the search for novel antitumor agents with distinct mechanisms of action. Ansamitocin P 3 attracted attention for its potent cytotoxicity against cancer cells and its unique mode of action involving inhibition of tubulin polymerization. Ansamitocin P 3 and its derivatives, such as maytansine and DM4, have been extensively studied for their potential in cancer therapy. These compounds serve as payloads for antibody-drug conjugates (ADCs), enabling targeted delivery of cytotoxic agents to cancer cells while sparing healthy tissues. Notably, ansamitocin P 3 is utilized in the ADC ado-trastuzumab emtansine (T-DM1) for the treatment of HER2-positive breast cancer. T-DM1 has demonstrated efficacy in patients who have progressed on prior HER2-targeted therapies and cytotoxic chemotherapy, offering a promising option for advanced disease. The success of T-DM1 underscores the therapeutic potential of ansamitocin P 3-based ADCs in oncology. Ongoing research aims to optimize ADC design, enhance tumor specificity, and broaden the utility of ansamitocin P 3 derivatives in the treatment of various malignancies, further solidifying their role in cancer therapy. References 2023. Biosynthesis of ansamitocin P-3 incurs stress on the producing strain Actinosynnema pretiosum at multiple targets. Communications Biology, 6(1). DOI: 10.1038/s42003-023-05227-w 2015. Improvement of Ansamitocin P-3 Production by Actinosynnema mirum with Fructose as the Sole Carbon Source. Applied Biochemistry and Biotechnology, 175(2). DOI: 10.1007/s12010-014-1445-6 1982. Biosynthetic origin of aminobenzenoid nucleus (C7N-unit) of ansamitocin, a group of novel maytansinoid antibiotics. The Journal of Antibiotics, 35(10). DOI: 10.7164/antibiotics.35.1415 |
| Market Analysis Reports |