Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Indoles |
|---|---|
| Name | 2-(2,6-Dioxopiperidin-3-yl)-4-fluoro-2,3-dihydro-1H-isoindole-1,3-dione |
| Synonyms | 2-(2,6-Dioxopiperidin-3-yl)-4-fluoroisoindolin-1,3-dione; 2-(2,6-Dioxopiperidin-3-yl)-4-fluoroisoindoline-1,3-dione; 4-Fluoro-thalidomide |
| Molecular Structure | 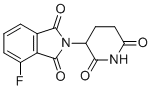 |
| Molecular Formula | C13H9FN2O4 |
| Molecular Weight | 276.22 |
| CAS Registry Number | 835616-60-9 |
| EC Number | 830-714-5 |
| SMILES | C1CC(=O)NC(=O)C1N2C(=O)C3=C(C2=O)C(=CC=C3)F |
| Solubility | Very slightly soluble (0.47 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.570±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2019 ACD/Labs) |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335-H361 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
The discovery of 2-(2,6-dioxopiperidin-3-yl)-4-fluoro-2,3-dihydro-1H-isoindole-1,3-dione stems from the study of biologically active piperidine and isoindole derivatives. Researchers have attempted to integrate fluorine atoms into the isoindole skeleton and dioxopiperidin moiety to create new compounds with enhanced therapeutic potential, hoping that it will provide better pharmacokinetic properties and biological efficacy. The synthesis of this compound involves a multi-step process combining fluorination, cyclization, and condensation reactions. Common methods include: cyclization to form the isoindole ring structure; fluorination to introduce fluorine atoms at the 4-position of the isoindole ring; condensation to couple with the dioxopiperidinyl group. The process produces the target compound with the desired purity and structural integrity, which can be characterized by methods such as NMR, IR, and mass spectrometry. The product has a 4-fluoroisoindole core and a 2,6-dioxopiperidinyl group, usually in the form of a crystalline solid or powder, soluble in organic solvents and slightly soluble in water. 2-(2,6-dioxopiperidin-3-yl)-4-fluoro-2,3-dihydro-1H-isoindole-1,3-dione shows promise in pharmaceutical applications due to its structural features: it exhibits cytotoxic activity against certain cancer cell lines. The unique structure of the compound favors interaction with biological targets, potentially inhibiting the growth and proliferation of cancer cells. Studies have found that the compound has the ability to inhibit viral replication. The presence of fluorine atoms and the dioxopiperidin moiety enhances its interaction with viral enzymes, making it a candidate for antiviral drug development. Studies have shown that the compound has potential use as an anti-inflammatory agent, and its ability to modulate inflammatory pathways may allow it to treat inflammatory diseases. In chemical research, the compound serves as a useful scaffold for the development of new derivatives and analogs. Its structure allows for modification, resulting in the synthesis of novel compounds with a variety of biological activities. Researchers use it to explore structure-activity relationships and optimize therapeutic properties. The compound is valuable in medicinal chemistry as a lead compound for drug discovery. Its synthesis and subsequent testing provide insights into how modifications of the piperidine or isoindole rings affect biological activity. This understanding guides the design of new drugs with improved efficacy and safety. As with many synthetic compounds, appropriate safety measures are necessary. Protective equipment such as gloves and goggles should be used when handling the compound to prevent contact. Proper storage in a cool, dry place is also essential to maintain its stability and prevent degradation. References 2023. Degradation of neurodegenerative disease-associated TDP-43 aggregates and oligomers via a proteolysis-targeting chimera. Journal of Biomedical Science, 30(1). DOI: 10.1186/s12929-023-00921-7 2022. A proteolysis-targeting chimera molecule selectively degrades ENL and inhibits malignant gene expression and tumor growth. Journal of Hematology & Oncology, 15(1). DOI: 10.1186/s13045-022-01258-8 2020. Targeted degradation of anaplastic lymphoma kinase by gold nanoparticle-based multi-headed proteolysis targeting chimeras. Colloids and surfaces. B, Biointerfaces, 188. DOI: 10.1016/j.colsurfb.2020.110795 |
| Market Analysis Reports |