Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai CR Corporation Limited | China | |||
|---|---|---|---|---|
 | www.crcorporation.cn | |||
 | +86 (21) 5736-6208 +86 13917420128 | |||
 | +86 (21) 5736-6228 | |||
 | fred.wen@crcorporation.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Nata Biochemical Products Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.natachem.com | |||
 | +86 (512) 6252-0998 | |||
 | +86 (512) 6252-7116 | |||
 | natamarket@natachem.com | |||
| Chemical manufacturer since 2000 | ||||
| Advanced Chemical Intermediates Ltd. | UK | |||
|---|---|---|---|---|
 | www.acints.com | |||
 | +44 (1840) 261-451 | |||
 | +44 (1840) 261-451 | |||
 | enquiries@acints.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Cyanopyridine |
|---|---|
| Name | 2,6-Dichloro-4-methylnicotinonitrile |
| Synonyms | 3-Cyano-4-methyl-2,6-dichloropyridine |
| Molecular Structure | 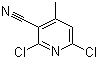 |
| Molecular Formula | C7H4Cl2N2 |
| Molecular Weight | 187.03 |
| CAS Registry Number | 875-35-4 |
| EC Number | 212-873-7 |
| SMILES | CC1=CC(=NC(=C1C#N)Cl)Cl |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 108-112 °C (Expl.) |
| Index of Refraction | 1.574, Calc.* |
| Boiling Point | 317.1±37.0 °C (760 mmHg), Calc.* |
| Flash Point | 145.5±26.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H312-H315-H317-H318-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
2,6-Dichloro-4-methylnicotinonitrile is an organic compound that belongs to the class of substituted nicotinonitriles. This compound contains a pyridine ring with two chlorine atoms at the 2 and 6 positions, a methyl group at the 4-position, and a nitrile group (-CN) attached at the 3-position of the pyridine ring. Its structure confers both chemical reactivity and specificity for use in various industrial applications. Known for its stability and functional properties, 2,6-dichloro-4-methylnicotinonitrile plays an important role in the synthesis of more complex chemical molecules and has applications in both pharmaceutical chemistry and material science. The synthesis of 2,6-dichloro-4-methylnicotinonitrile typically involves the halogenation of 4-methylpyridine to introduce chlorine atoms at the desired positions. The nitrile group is then introduced via a nucleophilic substitution reaction using cyanide, often facilitated by a catalyst to ensure high selectivity and yield. This method of synthesis allows for the precise control of functional groups, resulting in a highly reactive intermediate compound that can be further modified to suit specific applications. One of the major applications of 2,6-dichloro-4-methylnicotinonitrile lies in its use as a key intermediate in the synthesis of bioactive molecules, particularly in medicinal chemistry. The presence of the chloro and nitrile groups on the pyridine ring makes this compound a versatile scaffold for the development of drugs with specific biological activities. Its ability to act as a starting point for the creation of pharmacologically active derivatives is valuable in the search for new therapeutic agents, particularly for conditions such as cancer, inflammation, and infections. The nitrile group can participate in various chemical transformations, enabling the development of compounds with improved pharmacokinetic properties. In addition to its role in medicinal chemistry, 2,6-dichloro-4-methylnicotinonitrile has applications in the synthesis of agrochemicals. The compound serves as a precursor in the development of herbicides and pesticides due to the structural features that confer biological activity. The halogenated pyridine ring and nitrile functionality are common in many agrochemical compounds that exhibit selective activity against pests and weeds. By modifying the structure of 2,6-dichloro-4-methylnicotinonitrile, it is possible to design more efficient and environmentally friendly agrochemicals that can target specific pests or plant diseases. Moreover, the compound has been explored for use in material science, particularly in the development of functionalized polymers and coatings. The nitrile group provides reactivity that can be used in polymerization reactions, while the chloro and methyl substituents help modulate the physical properties of the resulting materials. 2,6-Dichloro-4-methylnicotinonitrile and its derivatives can be incorporated into conductive polymers, which are useful in the creation of electronic devices, sensors, and other advanced materials. The versatility of this compound in forming functional materials with tailored properties makes it a promising candidate for various technological innovations. In conclusion, 2,6-dichloro-4-methylnicotinonitrile is a versatile chemical compound with significant applications in drug discovery, agrochemical development, and material science. Its unique structure, containing halogen and nitrile groups on a pyridine ring, makes it an excellent building block for the synthesis of biologically active compounds, as well as functional materials. As research progresses, the applications of this compound are likely to expand, contributing to advancements in both industrial chemistry and high-performance materials. References 2021. Unusual regioselective reaction of 2,6-dichloro-4-methylnicotinonitrile with malononitrile dimer. Russian Chemical Bulletin, 70(7). DOI: 10.1007/s11172-021-3224-1 2018. Synthesis, structure, and biological activity of 2,6-diazido-4-methylnicotinonitrile derivatives. Chemistry of Heterocyclic Compounds, 54(10). DOI: 10.1007/s10593-018-2381-0 2018. The Guareschi-Thorpe Cyclization Revisited - An Efficient Synthesis of Substituted 2,6-Dihydroxypyridines and 2,6-Dichloropyridines. Synlett, 29(9). DOI: 10.1055/s-0037-1609685 |
| Market Analysis Reports |