Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Weinan Changfeng Science & Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cfkjwn.com | |||
 | +86 (913) 210-3869 | |||
 | +86 (913) 210-3869 | |||
 | 3303502436@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Beijing Guofang Dingsheng Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gfdschem.com | |||
 | +86 (10) 8904-9254 5909-2859 +86 18610472091 +86 18031679351 | |||
 | +86 (10) 5909-2859 | |||
 | gfdskj@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Kangchi Chemical Industry (shenzhen) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.konchie.com | |||
 | +86 (755) 8257-4253 | |||
 | +86 (755) 3315-1032 | |||
 | kangchi@konchie.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Xi'an Tuochao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xatuochao.cn | |||
 | +86 18092957403 | |||
 | chen.zeshan@xatuochao.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: +8618092957403 | |||
 | WhatsApp:+8618092957403 | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic palladium |
|---|---|
| Name | Bis(di-tert-butyl(4-dimethylaminophenyl)phosphine)dichloropalladium (II) |
| Synonyms | (SP-4-1)-Palladium dichloro-bis[4-[bis(1,1-dimethylethyl)phosphino-P]-N,N-dimethylbenzenamine]; PdCl2(Amphos)2 |
| Molecular Structure | 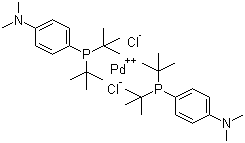 |
| Molecular Formula | C32H56Cl2N2P2Pd |
| Molecular Weight | 708.08 |
| CAS Registry Number | 887919-35-9 |
| EC Number | 627-800-8 |
| SMILES | CC(C)(C)P(C1=CC=C(C=C1)N(C)C)C(C)(C)C.CC(C)(C)P(C1=CC=C(C=C1)N(C)C)C(C)(C)C.Cl[Pd]Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Bis(di-tert-butyl(4-dimethylaminophenyl)phosphine)dichloropalladium(II), commonly abbreviated as [(dtbpf)PdCl₂], is a composite palladium catalyst that was developed following the development of palladium complex catalysis. It was synthesized in the early 21st century as a result of efforts to improve the efficiency and selectivity of palladium-catalyzed reactions. Its structural features are a palladium center coordinated to two chlorine atoms and two di-tert-butyl(4-dimethylaminophenyl)phosphine ligands. It has the appearance of a yellow crystalline solid that decomposes above 200°C. It is soluble in organic solvents such as dichloromethane, toluene, and tetrahydrofuran. [(dtbpf)PdCl₂] is widely used in the Suzuki-Miyaura coupling reaction to promote the formation of carbon-carbon bonds between aryl halides and boronic acids. Its bulky ligands enhance the stability and selectivity of the catalyst, allowing it to efficiently generate biaryls. It is also used in the Heck reaction to couple aryl halides with olefins to form substituted olefins. Its unique ligands provide steric protection, improve reaction efficiency and prevent side reactions. When used in Buchwald-Hartwig amination, [(dtbpf)PdCl₂] promotes the coupling of aryl halides with amines to form aromatic amines. The catalyst's structure promotes the formation of C-N bonds, which is essential for the production of pharmaceuticals and agrochemicals. The catalyst's role in forming carbon-carbon and carbon-nitrogen bonds makes it valuable in the synthesis of pharmaceutical intermediates, aiding in the development of complex drug molecules. Its applications extend to natural product synthesis, where precise and selective carbon-carbon couplings are required to build complex molecular frameworks. [(dtbpf)PdCl₂] modifies polymers and surfaces through coupling reactions, imparting new functional properties to materials. Its ability to promote carbon-carbon bond formation allows for the introduction of a variety of functionalities on the polymer backbone. In the electronics industry, the catalyst aids in the synthesis of materials for organic light-emitting diodes (OLEDs) and conductive polymers. Its efficiency in creating specific connections improves the performance of these materials in electronic applications. [(dtbpf)PdCl₂] is used to develop environmentally friendly catalytic processes. Its high activity and selectivity reduce the need for harsh reagents and conditions, promoting more environmentally friendly synthetic routes. The catalyst is used in the catalytic degradation process of environmental pollutants, promoting the decomposition of harmful compounds and aiding environmental cleanup efforts. The catalyst is used in academic research to explore the mechanisms of palladium-catalyzed reactions. Insights gained from these studies help design new catalysts and optimize existing catalysts for a variety of applications. [(dtbpf)PdCl₂] can serve as a model for the design of new palladium catalysts with enhanced properties, promoting innovation in the field of catalysis. References 2011. Synthesis of Unsymmetrical Arylheteroarylmethanes by Direct “On Water” Cross-Coupling between Benzylic and Heteroaromatic Halides. Chemistry - An Asian Journal, 6(6). DOI: 10.1002/asia.201100153 2011. Reaction with Arylboronic Acids (Suzuki and Suzuki-Miyaura Coupling). Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-116-00360 2010. Synthesis from Chloropyridazines or Chloropyridazinones via Palladium-Mediated Suzuki Coupling. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-116-00041 |
| Market Analysis Reports |