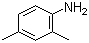
2,4-Dimethylaniline, an aromatic amine with the chemical formula C9H13N, consists of a benzene ring with two methyl groups and an amino group attached at the 2 and 4 positions. This compound was first synthesized in the late 19th century, reflecting the growing interest in the chemical modifications of aniline derivatives. The synthesis typically involves the alkylation of aniline with methyl groups, providing a straightforward route to obtain this compound.
The primary application of 2,4-dimethylaniline lies in the dye industry. It serves as a vital intermediate in the production of various azo dyes, which are widely used in textiles, plastics, and food industries due to their vibrant colors and stability. The ability of 2,4-dimethylaniline to undergo diazotization reactions allows it to be transformed into a variety of azo compounds, thus making it an essential building block in dye chemistry.
Beyond its role in dye synthesis, 2,4-dimethylaniline has applications in the formulation of agrochemicals. It is utilized in the production of herbicides and pesticides, contributing to the development of compounds that help manage agricultural pests and enhance crop yields. The effectiveness of 2,4-dimethylaniline-derived formulations in protecting plants highlights its significance in modern agriculture.
In addition to its industrial applications, 2,4-dimethylaniline is employed in the manufacture of rubber chemicals. It acts as an accelerator in rubber vulcanization, improving the processing and performance characteristics of rubber products. This application is particularly crucial in the automotive industry, where durable and high-performance rubber components are essential.
Despite its beneficial applications, safety concerns associated with 2,4-dimethylaniline warrant attention. As with many aromatic amines, it is recognized for its potential health risks, including carcinogenicity. Consequently, regulatory bodies have established guidelines for its safe handling and usage in industrial processes. Adequate safety measures, including personal protective equipment and proper ventilation, are essential when working with this compound to minimize exposure and health risks.
Recent research has begun to address the environmental impact of 2,4-dimethylaniline and its derivatives. There is a growing focus on developing more sustainable and environmentally friendly synthesis methods, as well as exploring alternative compounds that could serve similar functions in dye production and agricultural applications. This trend aligns with the broader movement towards sustainability in the chemical industry.
In summary, 2,4-dimethylaniline is an important compound with significant applications in dye synthesis, agrochemicals, and rubber manufacturing. Its discovery has contributed to advancements in these fields, although safety considerations are paramount. Ongoing research aims to enhance its applications while addressing environmental and health concerns associated with its use.
References
2022. Synthesis of Arylamines. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-236-00176
2022. Radical Cation Induced Synthesis of 2,3-Disubstituted Quinolines from Anilines and Aliphatic Aldehydes. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-115-00914
2022. Potassium Persulfate Mediated Synthesis of 4-Arylquinolines. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-115-01023
|















































 GHS06;GHS07;GHS08;GHS09 Danger Details
GHS06;GHS07;GHS08;GHS09 Danger Details