Online Database of Chemicals from Around the World
| Shenzhen Geneseqtools Bioscience & Technology Co. Ltd. | China | |||
|---|---|---|---|---|
 | geneseqtools.com | |||
 | +86 (0755) 2325-1735 | |||
 | sales@geneseqtools.com | |||
 | QQ Chat | |||
 | WeChat: 13302967066 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | (S)-5-Bromo-4-chloro-1H-indol-3-yl 2-aminopropanoate 2,2,2-trifluoroacetate |
| Synonyms | (5-bromo-4-chloro-1H-indol-3-yl) (2S)-2-aminopropanoate;2,2,2-trifluoroacetic acid |
| Molecular Structure | 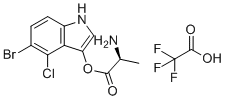 |
| Molecular Formula | C13H11BrClF3N2O4 |
| Molecular Weight | 431.59 |
| CAS Registry Number | 207725-18-6 |
| SMILES | C[C@@H](C(=O)OC1=CNC2=C1C(=C(C=C2)Br)Cl)N.C(=O)(C(F)(F)F)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
(S)-5-Bromo-4-chloro-1H-indol-3-yl 2-aminopropanoate 2,2,2-trifluoroacetate is a synthetic chemical compound that contains several distinct structural motifs, making it of interest in medicinal and biochemical research. The molecule comprises an indole core substituted with bromine at the 5-position and chlorine at the 4-position, a substitution pattern that is often associated with modulation of electronic and steric properties, potentially influencing biological activity. The indole nucleus is esterified at the 3-position with an L-alanine derivative ((S)-2-aminopropanoate), introducing chirality and enhancing its compatibility with biological systems. The compound is typically isolated or used as a salt with 2,2,2-trifluoroacetic acid, forming the trifluoroacetate salt, which increases solubility and handling stability in laboratory and clinical formulations. The discovery of this compound is rooted in the broader context of research on indole derivatives as pharmacologically active agents. Indoles, as a structural class, are widely present in nature and synthetic compounds with activities ranging from anticancer to antimicrobial to anti-inflammatory. The halogenated indole system in this compound—particularly the bromine and chlorine substitutions—has been studied for its ability to influence receptor binding and metabolic stability in drug discovery efforts. The presence of halogens on the indole ring can significantly affect the molecule’s lipophilicity, reactivity, and potential interactions with biological targets such as enzymes or receptors. One area of application for (S)-5-bromo-4-chloro-1H-indol-3-yl 2-aminopropanoate is as an intermediate or probe molecule in the synthesis or study of tryptophan analogues. Because it contains the basic scaffold of tryptophan modified with halogen substituents and esterified with an L-alanine moiety, it serves as a model compound in structure-activity relationship (SAR) studies aimed at understanding how variations in side-chain and ring substitutions affect the interaction of tryptophan-derived compounds with biological macromolecules. Such analogues are particularly relevant in the design of ligands for G-protein coupled receptors (GPCRs), serotonin receptors, and enzymes like indoleamine 2,3-dioxygenase (IDO), where modified tryptophan substrates can act as inhibitors or regulatory agents. The trifluoroacetate form of the compound is often chosen for preparative and biochemical applications due to the beneficial properties conferred by trifluoroacetic acid (TFA) as a counterion. TFA enhances the crystallinity, solubility in organic solvents, and shelf-life of amine-containing compounds. It is also volatile, which allows for easy removal under reduced pressure when needed during purification steps. In terms of synthetic strategy, the esterification of the indole-3-carboxylic acid with (S)-2-aminopropanol or the coupling of an activated carboxylic acid derivative with the chiral amino alcohol are common methods employed to construct this molecule. Protecting group strategies are often utilized during synthesis to ensure selectivity, particularly when dealing with multifunctional groups such as amines and phenols on the indole ring. Although not a clinically approved drug, this compound is valuable in research settings, especially where modified indole derivatives are used to investigate biological pathways or as starting materials for the synthesis of more complex drug candidates. The stereochemistry, halogenation pattern, and derivatization with amino acid-like moieties make it a versatile scaffold in pharmaceutical chemistry and chemical biology research. References none |
| Market Analysis Reports |