Online Database of Chemicals from Around the World
| Shanghai Rui Yun Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rychemical.com.cn | |||
 | +86 (21) 6726-7633 | |||
 | +86 (21) 6726-7655 | |||
 | sales@rychemical.com.cn | |||
 | WeChat: 13917251563 | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Analytical chemistry >> Standard >> Spectrometric standard |
|---|---|
| Name | Dimethyl (1-diazo-2-oxopropyl)phosphonate |
| Synonyms | 1-diazo-1-dimethoxyphosphorylpropan-2-one |
| Molecular Structure | 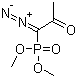 |
| Molecular Formula | C5H9N2O4P |
| Molecular Weight | 192.11 |
| CAS Registry Number | 90965-06-3 |
| EC Number | 692-521-0 |
| SMILES | CC(=O)C(=[N+]=[N-])P(=O)(OC)OC |
| Density | 0.800-0.850 g/mL (20 °C) |
|---|---|
| Refractive index | n20/D 1.352-1.354 |
| Hazard Symbols |    GHS02;GHS06;GHS07 Danger Details GHS02;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H301-H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P270-P271-P280-P301+P316-P301+P317-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Dimethyl (1-diazo-2-oxopropyl)phosphonate, also known as DM-DOPP, is an important compound in organic chemistry with notable applications in the synthesis of complex molecules. This compound contains both a diazo group and a phosphonate ester, making it a versatile reagent for various chemical transformations. The discovery of Dimethyl (1-diazo-2-oxopropyl)phosphonate is rooted in the broader exploration of diazo compounds and their applications in organic synthesis. Diazo compounds, characterized by the functional group -N2+, have been known for their ability to participate in a range of reactions, including cycloadditions and insertions. The combination of a diazo group with a phosphonate ester in DM-DOPP provides unique reactivity, making it a valuable tool for chemists. DM-DOPP is primarily used as a diazo transfer reagent, where it transfers the diazo group to other compounds. This process is particularly useful in the synthesis of diazo compounds from ketones or aldehydes, which can then be utilized in various reactions. The diazo group in DM-DOPP is highly reactive and can form stable intermediates that are useful in further synthetic steps. This property makes DM-DOPP a key reagent in the synthesis of complex molecules, including pharmaceuticals and natural products. One of the significant applications of DM-DOPP is in the formation of diazo compounds, which are intermediate species in organic synthesis. These diazo compounds can undergo a variety of reactions, such as cyclopropanation, carbene insertion, and rearrangements. The ability of DM-DOPP to generate diazo intermediates allows for the construction of complex structures with high precision. For example, in the synthesis of pharmaceuticals, diazo intermediates can be used to introduce functional groups into molecules, leading to the development of new drug candidates. Another notable application of DM-DOPP is in the preparation of phosphonate-containing compounds. Phosphonates are important in medicinal chemistry due to their ability to mimic phosphate groups and interact with biological systems. The presence of the phosphonate group in DM-DOPP facilitates the introduction of phosphonate moieties into various substrates, enabling the synthesis of phosphonate derivatives with potential biological activity. In addition to its applications in organic synthesis, DM-DOPP is also used in the development of new catalytic systems. The diazo group in DM-DOPP can be employed to generate reactive intermediates that participate in catalytic processes. This application is particularly relevant in the field of organocatalysis, where DM-DOPP can be used to create catalysts with unique properties for specific transformations. Overall, Dimethyl (1-diazo-2-oxopropyl)phosphonate is a versatile and valuable reagent in organic chemistry. Its ability to transfer diazo groups and introduce phosphonate moieties into molecules makes it an essential tool for the synthesis of complex compounds and the development of new catalytic systems. References 2016. Synthesis of Substituted 1,2,3-Triazoles via Metal-Free Click Cycloaddition Reactions and Alternative Cyclization Methods. Synthesis, 49(4). DOI: 10.1055/s-0036-1588904 2009. Bestmann-Ohira Reagent: A Versatile Reagent in Organic Synthesis. Synlett, 2009(16). DOI: 10.1055/s-0029-1218101 2021. Seyferth-Gilbert Homologation. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-235-00044 |
| Market Analysis Reports |