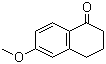
6-Methoxytetralone is an organic compound belonging to the tetralone family, characterized by a fused benzocycloalkanone structure with a methoxy functional group at the sixth position. This compound has gained attention in organic synthesis and medicinal chemistry due to its role as a key intermediate in the development of biologically active molecules.
The discovery of 6-methoxytetralone can be traced to research on substituted tetralones, which are widely used as building blocks in pharmaceutical and material sciences. The introduction of a methoxy group in the tetralone framework influences its electronic properties, making it valuable for targeted modifications in synthetic chemistry. The compound has been synthesized through various methods, including selective O-methylation of hydroxy-substituted tetralones and cyclization of aryl ketone precursors.
One of the primary applications of 6-methoxytetralone is in drug discovery, where it serves as a core scaffold in the synthesis of bioactive molecules, particularly in the development of neurological and anti-inflammatory agents. Its structural resemblance to natural and synthetic bioactive compounds enables researchers to explore its derivatives for potential therapeutic applications. Some derivatives have demonstrated promising activity against neurodegenerative disorders, owing to their interaction with key enzymatic pathways.
Additionally, 6-methoxytetralone has applications in material science, where its rigid bicyclic system contributes to the design of functionalized materials with improved stability. It has been explored in the synthesis of organic semiconductors and polymeric systems, where its electronic and steric properties play a crucial role in enhancing material performance.
In synthetic chemistry, 6-methoxytetralone is widely used as an intermediate in the preparation of complex heterocyclic compounds. Its reactivity allows for modifications at different positions, enabling the generation of structurally diverse compounds. This versatility makes it a valuable starting material for the synthesis of ligands, dyes, and other fine chemicals.
Ongoing research aims to expand the applications of 6-methoxytetralone in medicinal and industrial chemistry. Studies are focused on developing novel derivatives with enhanced pharmacological properties and exploring new methodologies for its efficient and sustainable synthesis. Given its structural significance and functional versatility, 6-methoxytetralone continues to be an important compound in modern chemical research.
References
1979. Oxidation of 6-methoxytetrahydronaphthalene in the presence of polyazopolyarenes. Bulletin of the Academy of Sciences of the USSR, Division of chemical science.
DOI: 10.1007/bf00963311
2015. Synthesis of an Intermediate of Nafoxidine via Nickel-Catalyzed Ketone Arylation. Synthesis.
DOI: 10.1055/s-0034-1380813
2022. A simple method for the rapid synthesis of 2-amino-7,7-dimethyl-5-oxo-1,4-diaryl-hexahydroquinoline-3-carboxamide derivatives. Journal of the Iranian Chemical Society.
DOI: 10.1007/s13738-020-01920-3
|












































 GHS07 Warning Details
GHS07 Warning Details