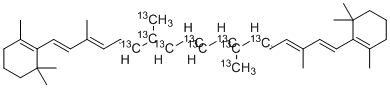(12,12′,13,13′,14,14′,15,15′,20,20′-13C10)-β,β-carotene is a stable isotopically labelled carotenoid analogue in which ten carbon atoms within the β-carotene molecule are enriched with the C isotope (ten C atoms distributed at carbons 12,12′,13,13′,14,14′,15,15′,20,20′). The molecule retains the core β,β-carotene structure (C40H56) featuring two β-ionone rings and an extended conjugated polyene chain, making it a valuable tracer for metabolic, nutritional or bioavailability studies of carotenoids in plants or humans. Because the C-labelling enables precise tracking by mass spectrometry or NMR, the compound is used in research to quantify uptake, conversion, tissue distribution or biotransformation of β-carotene with minimal isotopic interference from endogenous compounds.
In terms of synthesis and development, isotopically-labelled carotenoids such as C-β-carotene are produced using chemical or biosynthetic methods in which C-enriched starting materials (for example C-acetate, C-pyruvate, or C-isoprenoid units) are incorporated through the mevalonate/IPP pathway and carotenoid biosynthesis in engineered microorganisms such as *Escherichia coli*, *Pantoea ananatis* or *Dunaliella salina*. After accumulation of C-β-carotene, purification by chromatography yields the enriched compound which may then be formulated for human or plant tracer studies. The use of ten C atoms allows a distinct mass shift (for example +10 Da) compared with natural β-carotene (for example m/z 536) enabling sensitive quantitation in biological matrices.
Applications of (12,12′,13,13′,14,14′,15,15′,20,20′-C10)-β,β-carotene include metabolic fate studies in humans, animals and plants. In human nutritional research, this tracer is administered at dietary-relevant doses and blood/urine/faecal samples are analysed by LC–MS/MS or GC–MS to determine absorption, conversion to vitamin A, distribution to tissues (such as liver or adipose tissue), and excretion kinetics. In plant science, the compound facilitates studies of carotenoid turnover, photooxidation, conversion into downstream xanthophylls, and flux through the isoprenoid pathway. Because natural β-carotene isotopologues are abundant and endogenous levels high, the labelled variant provides a distinct analytical advantage for pharmacokinetic and metabolic modelling.
From an analytical perspective, the presence of multiple C atoms leads to characteristic isotopic envelopes in mass spectra and enables the application of stable isotope dilution assays (SIDA) in carotenoid quantitation. For example, co-elution of native and C-labelled β-carotene allows accurate determination of recovery, isomerisation and degradation under processing or storage conditions. The compound also facilitates NMR studies of carotenoid orientation in model membranes, as C-enrichment enhances signal-to-noise and permits detailed investigation of ring-chain conformational dynamics and interactions with lipid bilayers.
Overall, (12,12′,13,13′,14,14′,15,15′,20,20′-C10)-β,β-carotene exemplifies the advanced use of isotopic labelling in carotenoid research. Its availability and application bridge fundamental biological investigation (nutrient absorption, metabolic fate) and applied industrial or nutritional science (bioavailability, functional food development). As interest in carotenoid health effects and metabolism continues to grow, stable isotope tracers such as this compound provide crucial analytical precision and enable mechanistic insight beyond what unlabeled compounds alone can deliver.
References
Johnson EJ (2002) The role of carotenoids in human health. Nutrition in Clinical Care 5(2) 56–65. DOI: 10.1046/j.1523-5408.2002.00004.x
El-Agamey A, Lowe GM, McGarvey DJ, Mortensen A, Phillip DM, Truscott TG & Young AJ (2004) Carotenoid radical chemistry and antioxidant/pro-oxidant properties. Archives of Biochemistry and Biophysics 430(1) 37–48. DOI: 10.1016/j.abb.2004.03.007
|