Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hangzhou Yanshan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yanshan-chem.com | |||
 | +86 (571) 8689-7279 8669-6867 | |||
 | +86 (571) 2805-0260 | |||
 | sales@yanshan-chem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wuhan CheMax Chemical Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tm-chem.com | |||
 | +86 (27) 8384-0107 | |||
 | +86 (27) 8342-1702 | |||
 | tianmai@tm-chem.com | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Sris Synthesis Pvt Ltd. | India | |||
|---|---|---|---|---|
 | www.srissynthesis.com | |||
 | +91 (998) 952-2441 | |||
 | +91 (900) 065-7441 | |||
 | chari@srissynthesis.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Jiangyin Trust-Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.trust-chem.com | |||
 | +86 (510) 8625-7670 8626-8020 8626-9706 | |||
 | +86 (510) 8626-8021 | |||
 | trust@hi2000.com | |||
| Chemical manufacturer since 1996 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic acid |
|---|---|
| Name | 2-Chloro-4-methylsulphonylbenzoic acid |
| Synonyms | 2-Chloro-4-(methylsulfonyl)benzoic acid |
| Molecular Structure | 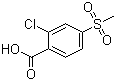 |
| Molecular Formula | C8H7ClO4S |
| Molecular Weight | 234.65 |
| CAS Registry Number | 53250-83-2 |
| EC Number | 406-520-8 |
| SMILES | CS(=O)(=O)C1=CC(=C(C=C1)C(=O)O)Cl |
| Melting point | 191-196 °C |
|---|---|
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H318 Details | ||||||||
| Safety Statements | P264+P265-P280-P305+P354+P338-aP317 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
2-Chloro-4-methylsulphonylbenzoic acid is an organic compound that has gained attention due to its role in various chemical and industrial applications. This compound, characterized by the presence of a chloro group, a methylsulfonyl group, and a carboxylic acid group attached to a benzene ring, offers unique chemical properties that make it useful in the synthesis of more complex molecules and in the development of specialized materials. The discovery of 2-Chloro-4-methylsulphonylbenzoic acid can be traced back to the systematic study of benzoic acid derivatives, which has been an area of significant interest in organic chemistry. Benzoic acid, a simple aromatic carboxylic acid, has long been a building block for the synthesis of a wide range of compounds. By introducing various functional groups, such as chloro and methylsulfonyl groups, chemists have been able to tailor the reactivity and properties of these derivatives for specific applications. One of the primary applications of 2-Chloro-4-methylsulphonylbenzoic acid is in organic synthesis. The compound's structure makes it a versatile intermediate in the synthesis of agrochemicals, pharmaceuticals, and specialty chemicals. The presence of the chloro group allows for nucleophilic substitution reactions, where the chlorine atom can be replaced by other functional groups, leading to the creation of more complex molecules. The methylsulfonyl group, known for its electron-withdrawing properties, enhances the reactivity of the benzene ring, making it more susceptible to various chemical transformations. This makes 2-Chloro-4-methylsulphonylbenzoic acid a valuable starting material in synthetic organic chemistry. In the agrochemical industry, derivatives of 2-Chloro-4-methylsulphonylbenzoic acid are used in the development of herbicides and pesticides. The compound's ability to be modified through substitution reactions allows for the creation of molecules that can selectively target specific biological pathways in plants or pests, leading to effective control of weeds or insect populations. These derivatives often work by inhibiting key enzymes or disrupting critical metabolic processes, making them powerful tools in modern agriculture. In the pharmaceutical industry, 2-Chloro-4-methylsulphonylbenzoic acid serves as an intermediate in the synthesis of drugs and active pharmaceutical ingredients (APIs). The compound's functional groups allow for further chemical modification, which is essential in the development of drug candidates with desired therapeutic properties. For example, the carboxylic acid group can be used to form esters or amides, which are common motifs in drug molecules. Additionally, the chloro and methylsulfonyl groups can be retained or replaced to fine-tune the pharmacokinetic and pharmacodynamic properties of the final drug molecule. Moreover, 2-Chloro-4-methylsulphonylbenzoic acid finds application in materials science. The compound's ability to participate in polymerization reactions makes it a useful monomer in the synthesis of specialized polymers. These polymers can be designed to have specific mechanical, thermal, or chemical properties, making them suitable for use in coatings, adhesives, and other advanced materials. The electron-withdrawing nature of the methylsulfonyl group also contributes to the stability and durability of the resulting materials. In summary, 2-Chloro-4-methylsulphonylbenzoic acid is a versatile chemical compound with significant applications in organic synthesis, agrochemicals, pharmaceuticals, and materials science. Its discovery as a derivative of benzoic acid has led to its use in the creation of more complex molecules and specialized materials, highlighting its importance in both scientific research and industrial applications. References 1998. Sulcotrione Soil Metabolism in Summer Corn Crops. Bulletin of Environmental Contamination and Toxicology, 61(5). DOI: 10.1007/s001289900813 2019. New relevant pesticide transformation products in groundwater detected using target and suspect screening for agricultural and urban micropollutants with LC-HRMS. Water Research, 165. DOI: 10.1016/j.watres.2019.114972 2024. Tandem catalysis enables chlorine-containing waste as chlorination reagents. Nature Chemistry, 16(5). DOI: 10.1038/s41557-024-01462-8 |
| Market Analysis Reports |