Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Amino alcohol derivative |
|---|---|
| Name | Tris(2-(2-methoxyethoxy)ethyl)amine |
| Synonyms | TDA-1; Tris(dioxa-3,6-heptyl)amine |
| Molecular Structure | 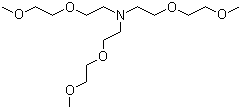 |
| Molecular Formula | C15H33NO6 |
| Molecular Weight | 323.43 |
| CAS Registry Number | 70384-51-9 |
| EC Number | 274-590-5 |
| SMILES | COCCOCCN(CCOCCOC)CCOCCOC |
| Density | 1.0±0.1 g/cm3, Calc.*, 1.008 g/mL (20 °C) (Expl.) |
|---|---|
| Index of Refraction | 1.446, Calc.*, 1.4486 (Expl.) |
| Boiling Point | 373.7±37.0 °C (760 mmHg), Calc.*, 163-171 °C (1 mm) (Expl.) |
| Flash Point | 98.6±23.7 °C, Calc.*, 162 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  HGS05 Danger Details HGS05 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H318 Details | ||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 2735 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Tris(2-(2-methoxyethoxy)ethyl)amine, also known as TMEEDA, is a compound with a variety of applications in industrial processes and materials science. Tris(2-(2-methoxyethoxy)ethyl)amine was synthesized and developed as a specialty amine compound to meet specific functional requirements in a variety of applications. Its chemical structure consists of three amine groups, each attached to a central carbon atom, which in turn is attached to three 2-(2-methoxyethoxy)ethyl groups. This molecular structure gives it unique properties that make it valuable in industrial processes and material formulations. TMEEDA is typically a clear to slightly yellow liquid that is soluble in water and a variety of organic solvents. Its functional groups include three amino (-NH₂) groups and three 2-(2-methoxyethoxy)ethyl groups. Tris(2-(2-methoxyethoxy)ethyl)amine can be synthesized by a multi-step organic synthesis process that involves reacting suitable starting materials under controlled conditions. The synthesis typically begins with the reaction of ethylene glycol with ethylene oxide to form 2-(2-methoxyethoxy)ethanol, which is then further reacted to introduce amine groups. TMEEDA is used as an ingredient in polymer formulations, particularly in polyurethane and epoxy systems. It acts as a chain extender and crosslinker, improving the flexibility, durability, and mechanical properties of polymers. It can be used as a corrosion inhibitor in metalworking fluids and coatings, helping to protect metal surfaces from degradation caused by exposure to corrosive environments. TMEEDA is used as a ligand in catalytic reactions and organometallic chemistry, promoting complex formation and catalytic activity in organic synthesis. It is used in electrolyte formulations for batteries and capacitors to help enhance ionic conductivity and stability. TMEEDA is used in surface treatment formulations to enhance adhesion and performance characteristics of coatings and adhesives. As with handling any compound, proper safety precautions should be observed when working with tris(2-(2-methoxyethoxy)ethyl)amine. This includes wearing appropriate personal protective equipment (PPE) and following recommended handling procedures to minimize exposure risk. Research and development efforts continue to explore new applications and formulations for TMEEDA. Advances in polymer science, materials engineering, and electrochemistry are expected to further expand its utility and effectiveness in a variety of industrial and technological fields. References 2021. Equilibrium States in Aqueous Solutions of Some Ionic Liquids. Surface Engineering and Applied Electrochemistry, 57(1). DOI: 10.3103/s1068375521010063 2011. Bifunctional Nanoparticles with Fluorescence and Magnetism via Surface-Initiated AGET ATRP Mediated by an Iron Catalyst. Langmuir : the ACS journal of surfaces and colloids, 27(18). DOI: 10.1021/la202749v 2009. Fe(III)-catalyzed AGET ATRP of styrene using triphenyl phosphine as ligand. Polymer Bulletin, 63(2). DOI: 10.1007/s00289-009-0139-7 |
| Market Analysis Reports |