Online Database of Chemicals from Around the World
|
CAS: 83708-10-5 Product: Hydroxynorfentanyl No suppilers available. |
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | Hydroxynorfentanyl |
| Synonyms | 3-hydroxy-N-phenyl-N-piperidin-4-ylpropanamide |
| Molecular Structure | 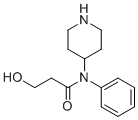 |
| Molecular Formula | C14H20N2O2 |
| Molecular Weight | 248.32 |
| CAS Registry Number | 83708-10-5 |
| SMILES | C1CNCCC1N(C2=CC=CC=C2)C(=O)CCO |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 415.5±40.0 ºC 760 mmHg (Calc.)* |
| Flash point | 205.1±27.3 ºC (Calc.)* |
| Index of refraction | 1.581 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
Hydroxynorfentanyl is a major phase I metabolite of fentanyl, a potent synthetic opioid widely used in clinical anesthesia and pain management. Structurally, hydroxynorfentanyl is derived from norfentanyl by the addition of a hydroxyl group, typically introduced via oxidative metabolism in the liver. It retains the core anilidopiperidine framework of fentanyl, but the modifications it undergoes during metabolism significantly reduce its pharmacological activity compared to the parent compound. The formation of hydroxynorfentanyl occurs primarily through the action of cytochrome P450 enzymes, particularly CYP3A4, which mediate the oxidative biotransformation of fentanyl. After initial N-dealkylation of fentanyl to form norfentanyl, further hydroxylation leads to the production of hydroxynorfentanyl. This metabolic route represents a detoxification process in the body, as both norfentanyl and hydroxynorfentanyl are considered inactive or significantly less active at the μ-opioid receptor. The presence of hydroxynorfentanyl in biological specimens, such as urine or plasma, is of considerable forensic and clinical interest. It serves as a biomarker for fentanyl exposure and metabolism. In toxicological investigations, particularly in cases of suspected fentanyl use, overdose, or poisoning, the detection of hydroxynorfentanyl can confirm prior fentanyl administration even when the parent compound is no longer detectable due to its rapid clearance. Its relative abundance and stability make it a preferred analyte for monitoring fentanyl usage in both therapeutic and illicit contexts. Analytical methods for detecting hydroxynorfentanyl typically involve liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). These methods provide high sensitivity and specificity, enabling accurate quantification in complex biological matrices. Standardized reference materials and calibration standards are essential for ensuring the reliability of such assays. Hydroxynorfentanyl has limited pharmacological relevance on its own, as it exhibits minimal binding to the μ-opioid receptor. Its lack of significant opioid activity has been confirmed in various in vitro and in vivo studies. However, its role as a non-active metabolite contributes to understanding fentanyl’s metabolic clearance and the time course of its pharmacological effects. In the broader context of opioid pharmacokinetics, the identification and characterization of metabolites like hydroxynorfentanyl are important for improving clinical safety. Understanding the metabolism of fentanyl helps guide dosing adjustments in populations with impaired hepatic function and assists in interpreting drug test results. It also provides insights into the potential for drug-drug interactions involving enzymes that may affect fentanyl clearance. In regulatory and workplace drug testing programs, hydroxynorfentanyl is frequently targeted in screening protocols. Its detection is often used to confirm fentanyl ingestion, particularly in cases where direct detection of fentanyl is hindered by its rapid metabolism or low concentrations. The inclusion of hydroxynorfentanyl in drug testing panels enhances the diagnostic window for identifying fentanyl use. Overall, hydroxynorfentanyl plays a critical role in the pharmacological and toxicological evaluation of fentanyl. While it lacks opioid activity, its formation reflects the body’s metabolic handling of fentanyl and provides a reliable marker for exposure. Its significance lies not in its effects, but in its utility for clinical monitoring, forensic investigation, and public health surveillance related to fentanyl use and misuse. |
| Market Analysis Reports |
| List of Reports Available for Hydroxynorfentanyl |