Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink massive supplier since 2021 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (510) 219-6317 | |||
 |
sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (551) 6541-8684 | |||
 |
sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2010 | ||||
| BOC Sciences | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (631) 485-4226 | |||
 |
info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2010 | ||||
| Suzhou Fosai New Material Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 18662206522 | |||
 |
sale_fosai@163.com zhou.xiaojin@fosai.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8771-1850 | |||
 |
market19@leapchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink standard supplier since 2015 | ||||
| Hunan Chemfish Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (731) 8556-7275 | |||
 |
sales@chemfish.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink standard supplier since 2018 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal fluorides and salts |
|---|---|
| Name | Lithium tetrafluoroborate |
| Molecular Structure | 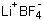 |
| Molecular Formula | LiBF4 |
| Molecular Weight | 93.74 |
| CAS Registry Number | 14283-07-9 |
| EC Number | 238-178-9 |
| SMILES | [Li+].[B-](F)(F)(F)F |
| Density | 0.852 g/mL (Expl.) |
|---|---|
| Melting point | 293 - 300 ºC (Decomposes) (Expl.) |
| Solubility | soluble in water (Expl.) |
| Hazard Symbols |

 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H302-H312-H314-H315-H318-H319-H332 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Precautionary Statements | P260-P261-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P321-P330-P332+P317-P337+P317-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3260 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lithium tetrafluoroborate is an inorganic salt with the chemical formula LiBF4. It consists of lithium cations and tetrafluoroborate anions and is typically obtained as a white crystalline solid that is soluble in water and in various polar organic solvents. The compound emerged from early 20th-century investigations into boron-fluorine chemistry, following the identification of boron trifluoride and its ability to form complex fluoroborate anions. Systematic studies of boron halides and their derivatives established the tetrafluoroborate ion as a stable, weakly coordinating anion, enabling the preparation and characterization of salts such as lithium tetrafluoroborate. The development of fluoroborate chemistry was closely linked to advances in inorganic fluorine chemistry and coordination theory. Researchers recognized that the tetrafluoroborate anion possesses a tetrahedral structure around boron, with strong boron-fluorine bonds contributing to its stability. The weakly coordinating nature of the anion makes it useful for stabilizing reactive cationic species without participating strongly in coordination. As a result, lithium tetrafluoroborate became an important reagent and supporting electrolyte in both laboratory and industrial contexts. One of the most significant applications of lithium tetrafluoroborate has been in electrochemistry. With the rise of nonaqueous electrochemical systems in the mid-20th century, salts capable of dissolving in organic solvents and providing high ionic conductivity were required. Lithium tetrafluoroborate was identified as a suitable supporting electrolyte because it dissociates effectively in aprotic solvents and exhibits good electrochemical stability over a relatively wide potential window. It has been employed in research on lithium batteries and other electrochemical devices, where its conductivity and compatibility with organic carbonate solvents have been systematically evaluated. In lithium battery technology, lithium salts serve as charge carriers within the electrolyte solution. Lithium tetrafluoroborate has been used in combination with solvents such as ethylene carbonate and dimethyl carbonate to formulate electrolytes for rechargeable lithium cells. Studies have documented its ionic conductivity, thermal behavior, and influence on electrode interfaces. Although other lithium salts have also been developed and widely adopted, lithium tetrafluoroborate remains a well-characterized alternative in specific battery formulations and in fundamental electrochemical investigations. Beyond battery systems, lithium tetrafluoroborate has found application in organic synthesis. The tetrafluoroborate anion is frequently used to generate stable salts of reactive cationic intermediates, including diazonium and carbocation species. Lithium tetrafluoroborate can participate in salt metathesis reactions, enabling the preparation of tetrafluoroborate salts of organic cations. Such salts are often more stable and less hygroscopic than corresponding halide salts, facilitating isolation and purification. In some cases, lithium tetrafluoroborate has also been employed as a Lewis acid source in catalytic transformations, owing to the presence of boron-fluorine bonds and its ability to interact with electron-rich substrates under controlled conditions. In materials science and surface treatment, fluoroborate salts have been utilized in electroplating and metal finishing processes. Lithium tetrafluoroborate solutions have been investigated as components of plating baths, where fluoroborate complexes can influence metal ion solubility and deposition behavior. The stability of the tetrafluoroborate anion under acidic conditions contributes to its usefulness in such environments. The physicochemical properties of lithium tetrafluoroborate, including its solubility, hygroscopicity, and thermal decomposition behavior, have been characterized by analytical techniques such as X-ray diffraction and thermal analysis. Upon heating to sufficiently high temperatures, it decomposes to release boron trifluoride and other fluorine-containing species. These well-documented properties inform its handling and storage in laboratory and industrial settings. From its origins in foundational studies of boron-fluorine compounds to its established roles in electrochemistry, organic synthesis, and materials processing, lithium tetrafluoroborate illustrates the practical importance of stable complex anions in modern chemistry. Its defined structure, reliable ionic behavior, and compatibility with diverse chemical systems have ensured its continued application in research and technology. References 2025. Review on environmental impacts of various types of power batteries using LCA. Environment, Development and Sustainability. DOI: 10.1007/s10668-025-06513-9 2025. On the Possibility of Estimating the Dissociation Constant of Lithium Salts in Sulfolane by Molecular Dynamics. Russian Journal of Physical Chemistry A. DOI: 10.1134/s0036024425701006 2025. Recent achievements on nonflammable triethyl phosphate-based electrolytes for stable and safe lithium metal batteries. Science China Chemistry. DOI: 10.1007/s11426-025-2679-3 |
| Market Analysis Reports |
| List of Reports Available for Lithium tetrafluoroborate |