Online Database of Chemicals from Around the World
| Taizhou Tongxin Biopharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyrise.com | |||
 | +86 18652728585 | |||
 | +86 (523) 8276-5215 | |||
 | sales@allyrise.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Qingdao Xinnuo Pharmaceutical Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinnuopharma.com | |||
 | +86 (531) 6656-1050 +86 18615688656 +86 18653156686 | |||
 | +86 (531) 6656-1050 | |||
 | sales@xinnuopharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Enki Biopharmaceuticals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.enkibiopharma.com | |||
 | +86 (21) 5768-0965 +86 13916707528 | |||
 | +86 (21) 5768-0922 | |||
 | info@enkibiopharma.com | |||
 | QQ Chat | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Zhuhai Hairuide Bioscience and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhhairuide.com | |||
 | +86 (756) 778-9199 +86 13326687259 +86 13326687289 +86 15913232160 | |||
 | +86 (756) 778-9099 | |||
 | sales@zhhairuide.com zhengyanhong@zhhairuide.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Axon MedChem BV | Netherlands | |||
|---|---|---|---|---|
 | www.axonmedchem.com | |||
 | +31 (50) 311-8007 | |||
 | +31 (50) 360-0390 | |||
 | order@axonmedchem.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Digestive system medication >> Acid and gastric mucosal protective drugs |
|---|---|
| Name | 5-(2-Fluorophenyl)-N-methyl-1-(3-pyridinylsulfonyl)-1H-pyrrole-3-methanamine (2E)-2-butenedioate |
| Synonyms | (E)-but-2-enedioic acid;1-[5-(2-fluorophenyl)-1-pyridin-3-ylsulfonylpyrrol-3-yl]-N-methylmethanamine |
| Molecular Structure | 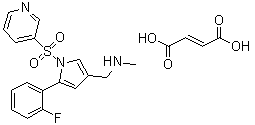 |
| Molecular Formula | C17H16FN3O2S.C4H4O4 |
| Molecular Weight | 461.46 |
| CAS Registry Number | 881681-01-2 |
| EC Number | 863-483-4 |
| SMILES | CNCC1=CN(C(=C1)C2=CC=CC=C2F)S(=O)(=O)C3=CN=CC=C3.C(=C/C(=O)O)C(=O)O |
| Solubility | 15 mg/mL (DMSO) |
|---|---|
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H373 Details | ||||||||||||||||
| Safety Statements | P260-P264-P270-P301+P317-P319-P330-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
5-(2-Fluorophenyl)-N-methyl-1-(3-pyridinylsulfonyl)-1H-pyrrole-3-methanamine (2E)-2-butenedioate, often referred to by its shorthand, was discovered during investigations aimed at enhancing the pharmacological profiles of pyrrole-based compounds. The synthesis involves a multi-step process starting with the derivatization of a pyrrole core and incorporating fluorophenyl and pyridinylsulfonyl groups, followed by quaternization and salt formation with fumaric acid ((2E)-2-butenedioate). This synthesis combines fluorination and sulfonylation techniques to enhance the compound's biological activity, stability, and solubility. The unique structure of the resulting compound offers diverse applications in medicinal chemistry. This compound has shown potential in the development of anticancer drugs. The combination of the fluorophenyl and pyridinylsulfonyl groups enhances its ability to interact with cancer cell targets, particularly kinases and DNA. This interaction can inhibit the growth of cancer cells by disrupting their signaling pathways. As a fumarate salt, it improves the solubility and bioavailability, making it a promising candidate for oral drug formulations. Its structure also lends itself to anti-inflammatory applications. By modulating pathways involving cytokines and inflammatory mediators, it can potentially reduce inflammation. This makes it a valuable scaffold for designing drugs for chronic inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease. The compound's ability to cross the blood-brain barrier due to its fluorophenyl moiety opens avenues for neurological applications. It can be explored for the treatment of neurodegenerative diseases like Alzheimer's or Parkinson's by targeting specific neural pathways and receptors. The presence of the pyridinylsulfonyl group allows the compound to act as an enzyme inhibitor. It can be utilized to inhibit enzymes involved in metabolic pathways or disease processes. This makes it a useful tool in studying enzyme functions and developing enzyme-targeted therapies. Its structure is suitable for studying protein-ligand interactions in drug discovery. By binding to active sites or allosteric sites of proteins, it can modulate protein functions, providing insights into protein behavior and aiding in the design of novel inhibitors or activators. The compound's unique chemical features make it useful as a molecular probe. It can be used to detect specific biological molecules or environmental contaminants. This is particularly useful in diagnostic applications where specific and sensitive detection is crucial. The conjugated system involving the pyrrole and fluorophenyl groups gives the compound interesting electronic properties, making it a candidate for use in organic semiconductors. It can be used in the development of electronic devices that require efficient charge transport, such as organic field-effect transistors (OFETs). As a ligand, this compound can form complexes with transition metals, which are useful in catalysis. These complexes can facilitate various chemical reactions by providing a suitable environment for the reactants, increasing reaction rates, and improving product yields. References 2012. Discovery of a Novel Pyrrole Derivative 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine Fumarate (TAK-438) as a Potassium-Competitive Acid Blocker (P-CAB). Journal of Medicinal Chemistry, 55(9). DOI: 10.1021/jm300318t 2010. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine Monofumarate (TAK-438), a Novel and Potent Potassium-Competitive Acid Blocker for the Treatment of Acid-Related Diseases. The Journal of Pharmacology and Experimental Therapeutics, 335(1). DOI: 10.1124/jpet.110.170274 2011. A Study Comparing the Antisecretory Effect of TAK-438, a Novel Potassium-Competitive Acid Blocker, with Lansoprazole in Animals. The Journal of Pharmacology and Experimental Therapeutics, 337(3). DOI: 10.1124/jpet.111.179556 |
| Market Analysis Reports |